Introduction
Cells of tissues and organs are constantly exposed to oxidative stress and free radicals every day. Normal cells respond by using antioxidant enzymes such as glutathione (GSH), vitamins C and E, catalase, superoxide dismutase. Sophisticated DNA repair mechanisms are also used1).
If the balance between the production of free radicals and the defense mechanisms of cells is broken, the human body is exposed to oxidative stress. Oxidative stress causes chronic diseases such as cancer, diabetes, neurodegenerative diseases and cardiovascular disease. To overcome this, many studies have been done on the antioxidant effects of natural products. Long-term intake of antioxidant-rich foods may delay or avoid the development of related diseases2).
According to Brewer’s research3), antioxidants are generally thought to be more superior to natural antioxidants than synthetic antioxidants because they are proportional to the number of hydroxyl (OH) groups present in the aromatic rings, therefore research continues to find new natural products with antioxidant properties.
Continuous exposure of cells to oxidative stress increases the expression of genes that cause degenerative diseases, leading to chronic inflammatory responses.
Inflammatory responses are involved in the production and regulation of pro-inflammatory cytokines such as nitric oxide, prostaglandins and TNF-α in macrophages. NO is an inflammatory mediator and is produced by nitric oxide synthases(NOS). Inducible nitric oxide synthase (iNOS) stimulates the inflammatory response by producing NO in macrophages involved in the inflammatory response4). Cyclooxygenase-2(COX-2) also stimulates prostaglandin biosynthesis as well as NO production. The inflammatory response also acts as a normal defense mechanism of the human body, but if the inflammatory response becomes chronic, it can cause inflammatory diseases such as rheumatoid arthritis, arteriosclerosis, gastritis and asthma5).
Paulownia tomentosa is a deciduous tree belonging to the paulownia family distributed in Korea, China, and Japan. It is about 15m high, its branches starve and spread, and the bark is grayish brown with hairs on young branches. Paulownia tomentosa can be used to treat deodorants, hair growth, gonorrhea and bruises6). Paulownia tomentosa is known to contain catapol, syringin, aucabin, caniferin, actcoside, paulownin, sesamin, (+)-pipertol and the like. Pharmacological studies have also reported antimicrobial activity against Staphlococcus aureus and Pseudomonas aeruginosa7,8).
Although methanol, ethanol and water soluble extracts of Paulownia tomentosa fruit were compared with antioxidant activity9), the antioxidant and anti-inflammatory effects of Paulownia tomentosa branches have not been studied.
In this study, the antioxidant and anti-inflammatory activity of Paulownia tomentosa was analyzed by using ethanol extract of Paulownia tomentosa, and the anti-inflammatory activity was analyzed through RAW264.7 macrophages induced by LPS.
Materials and methods
1. Reagent
The domestic paulownia tree used in this experiment was purchased from Samhong Construction Co., Ltd. (Seoul).
Reagents such as Folin-Ciocalteu’s reagent, ferric chloride (FeCl3), 1,1-diphenyl-2-picrylhydrazyl (DPPH) 2,2′-azino-bis(3-ethylbenzothiazoline-6- sulfonic acid)(ABTS), potassium persulfate, 4,6-tripyridyl-S-triazine (TPTZ) used in the experiment were Sigma-Aldrich Ltd. (St. Louis, MO, USA), all other reagents used were express.
2. Extraction of samples
The paulownia sample used in this experiment was extracted using ethanol.
1.0 g of ethanol was added to 100 g of a real paulownia sample, extracted three times at room temperature for 24 hours, filtered through filter paper(No. 41, Whatman, Maidstone, England), and concentrated by rotary vacuum evaporator (EYELA, Tokyo, Japan) and lyophilized.
Extracted paulownia samples were stored frozen and used in the experiment.
3. Total Polyphenol Content Determination
Total polyphenol content was determined by slightly modifying the method of Singleton and Rossi10).
To 200 μl of paulownia extract, add 1.0 N Folin-Ciocalteau Reagent and 20% Na2CO3 solution in 1.0 mL each. The reaction was carried out at room temperature for 30 minutes. And then, the absorbance was measured at 700 nm using a microplate spectrophotometer(Thermo Scientific Multiskan GO, Thermo Fisher Scientific, Ratastie, FINLAND).
Gallic acid was used as a standard to calculate the total polyphenol content of the paulownia extract and expressed as gallic acid equivalents (mg GAE/g extract).
4. Total Flavonoid Content Determination
Total flavonoid content slightly modified the method of Jia et al11).
1.25 mL of distilled water was mixed with 0.25 mL of paulownia extract and 75 μL of 5% sodium nitrite was added and allowed to react at room temperature for 5 minutes.
After adding 0.15 mL of 10% aluminum chloride and reacting for 6 minutes at room temperature, 0.5 mL of 1.0 M sodium hydroxide and 275 μL of distilled water were added and mixed in order, and then the absorbance was measured at 510 nm. Total flavonoid content of paulownia extract was calculated using catechin as standard and expressed as catechin equivalents (mg CE/g extract).
5. DPPH Radical Scavenging Activity by Electron Spin Resonance(ESR)
DPPH radical scavenging ability followed the measurements performed by Lee et al12). 60 μL dissolved in methanol and 60 μL of DPPH and 60 μL of various concentrations of the sample were mixed well and allowed to react at room temperature for 2 minutes. And then, It was transferred to a capillary tube and measured with an ESR spectrometer(Jeol Co.Ltd., Tokyo, Japan). The measurement conditions were central field: 3475 G, modulation frequency: 100 kHz, modulation amplitude: 2 G, microwave power: 5 mW, gain: 6.3 × 105, and temperature: 298 K.
6. Total Antioxidant Activity Using ABTS Radicals
Antioxidant activity using ABTS radical was slightly modified by the ABTS cation decolorization assay method13). 7.4 mM ABTS solution and 2.4 mM potassium persulfate were mixed and reacted in the dark for 12–16 hours to generate ABTS radicals. The absorbance of the generated ABTS radicals was diluted with distilled water to 1.5 at 734 nm, 1.0 mL was taken, 100 μL of the sample was added, and reacted at room temperature for 10 minutes to measure absorbance at 734 nm.
The results were expressed as mM Trolox equivalent/mg extract using Trolox as standard.
7. Determination of Total Antioxidant Capacity Using Ferric reducing antioxidant power (FRAP)
FRAP measurements were taken by Benzie and Strain14).
FRAP reagent was prepared by mixing 300 mM acetate buffer (pH 3.6), 10 mM TPTZ dissolved in 40 mM HCl, and 20 mM FeCl3.6H2O at a ratio of 10:1:1(v/v/v). 0.15 mL of sample and 3.0 mL of FRAP reagent were mixed and reacted at 37 °C for 5 minutes, and the absorbance was measured at 593 nm.
The results were expressed as mM FeSO4 equivalent/mg extract using FeSO4 as standard.
8. Cell culture
Macrophages (RAW264.7) were cultured in Dulbecco’s modified eagle medium (DMEM) medium containing 10% inactivated fetal bovine serum (FBS) and 1.0% penicillin-streptomycin at 37 °C and 5% CO2.
9. Cytotoxicity measurement
To evaluate the cytotoxicity of paulownia extract on macrophages, the survival rate of the cells was measured by slightly modifying the method of Berridge and Tan15).
RAW 264.7 cells were seeded in 96-well plates at a concentration of 2×104 cells/well and precultured for 20 hours. Then paulownia extract was added by concentration (25, 50, 100, 200 μ g/mL) and incubated for 24 hours.
After 2 hours after adding 0.2 mg/mL MTT solution, the supernatant was discarded, and 150 μL of dimethyl sulfoxide (DMSO) was added to dissolve the generated formazan, and the absorbance was measured at 540 nm.
Cell viability was calculated by the following method.
10. Nitric oxide production measurement
In order to confirm the anti-inflammatory effect of paulownia extract on the inflammatory response of LPS-induced RAW264.7 cells, the nitrite concentration in the culture was measured.
RAW264.7 cells were dispensed into 24-well plates at a concentration of 6×104 cells/well and incubated 20 hours before in a 5% CO2 incubator.
Then paulownia extract was treated with 0, 50, 100, 200 μg/mL. After one hour, 100 ng/mL of LPS was treated and then incubated for 18 hours.
The supernatant of the culture solution and Griess reagent were mixed 1: 1 and reacted at room temperature for 10 minutes, and the absorbance was measured at 540 nm using a microplate reader.
Sodium nitrite(NaNO2) was used as a standard curve to calculate the amount of NO produced in the cell culture.
11. Pro-inflammatory cytokines secretion measurement
The secretion of IL-6 and TNF-α cytokine in cell culture medium of RAW 264.7 cells was measured using an ELISA kit (BD Biosciences, San Diego, CA, USA). The method is as follows.
RAW264.7 cells were dispensed in 24-well plates at a concentration of 6×104 cells/well and precultured for 18 hours. Then paulownia extract was treated to a concentration of 50, 100, 200 μ g/mL and after one hour was treated with 100 ng/mL LPS. The supernatant was recovered after 18 hours of incubation. The IL-6, TNF-α capture antibody was diluted in the coating buffer on the microplate and coated overnight at 4°C.
After washing with PBS-T containing 0.05% Tween 20 and blocked with 10% FBS solution. After washing with PBS-T, the cell culture supernatant was added to each microplate well and reacted for 2 hours at room temperature.
After the reaction, washed with PBS-T, diluted biotinylated anti-mouse IL-6, TNF-α detection antibody was added and reacted for 1 hour. Then streptavidin horseradish peroxidase conjugate was added and reacted for 30 minutes. After the reaction, it was washed again with PBS-T and the reaction was carried out in the dark for 30 minutes at room temperature by the addition of Substrate solution. After the reaction was terminated with 2N H2SO4, the absorbance was measured at 450 nm using a microplate reader.
12. Inflammation-related protein expression analysis
To investigate the effect of paulownia extract on inflammation-related protein expression, RAW 264.7 cells were cultured in 6 well plates at a concentration of 1×106 cells/well and cultured 18 hours ago. Paulownia extract was pretreated for one hour and then treated with LPS.
After culturing, the cells were collected and washed with PBS (phosphate buffered saline), lysis buffer was added, lysis was performed at 4°C for 15 minutes, and the protein was recovered after centrifugation at 13,000 rpm for 15 minutes. Proteins were quantified by Bradford protein assay method, and the quantified proteins were separated by 10–12% SDS-PAGE in an amount of 15–20 μl. The isolated protein was transferred to a polyvinylidene difluoride (PVDF) membrane (GE Healthcare Life Science, Pittsburgh, PA, USA) for 2 hours, and blocked for 1 hour at room temperature with tris buffered saline with tween 20(TBS-T) solution containing 5% skim milk. Antibodies for examining the expression levels of iNOS, COX-2 and MAPK were diluted 1: 1,000 with anti-mouse iNOS, COX-2 and MAPK and reacted overnight in refrigeration and then washed three times with TBS-T.
As a secondary antibody, anti-mouse IgG and anti-rabbit IgG conjugated with HRP (horseradish peroxydase) were diluted 1: 1,000 and reacted at room temperature for 1 hour. After washing three times with TBS-T and reacting with ECL substrate for 1–3 minutes, each protein band was visualized using an image analyzer (Davinch-Western™, Youngwha Scientific Co., Seoul, Korea).
13. Nucleoprotein Extraction
In order to confirm the expression level of NF-κB p65, RAW264.7 cells were pretreated with paulownia extract for one hour, and then treated with LPS for 15 minutes, and then nuclear proteins were isolated.
Nuclear and cytoplasmic extraction was performed using a NE-PER Nuclear Protein extraction kit (Thermo Scientific, Rockford, IL, USA), and the protein was quantified by the Bradford protein assay method.
14. Statistical analysis
The experimental results were obtained by the mean ± standard deviation (SD) according to each item, and the significance of each group was verified using GraphPad Prism 5.0 version (GraphPad Software, La Jolla, CA, USA).
One-way ANOVA was performed, and post-test was carried out by Tukey’s and Dunnett’s test methods. It was determined that the P value was less than 0.05.
Results and Discussion
1. Extraction Yield and Total Polyphenols and Flavonoid Contents of Paulownia
As a result of measuring the yield of paulownia extract, it was 1.086% (Table 1). The results of total polyphenol and total flavonoid content, which influence the physiological activity of paulownia extract, are shown in Table 1.
The total polyphenol content of Paulownia extract was 143.98 ± 1.84 mg GAE/g extract and the total flavonoid content was 115.33 ± 4.16 mg CE/g extract.
2. DPPH radical scavenging activity using ESR
Table 2 shows the results of measuring DPPH radical scavenging activity of paulownia extract using ESR.
Paulownia extract showed IC50 value of 50% scavenging activity concentration of DPPH radical and showed high DPPH radical scavenging activity at 0.025 ± 0.001 mg/mL. The IC50 value of vitamin C used as a positive control was 0.003 ± 0.001 mg/mL, indicating lower radical scavenging activity than vitamin C.
3. Total Antioxidant Activity Using ABTS Radicals
The ABTS radical scavenging activity of Paulownia extract is shown in Table 3. Using Trolox as a standard, total antioxidant capacity using ABTS radicals was expressed as TEAC (mM Trolox eq./mg extract).
The TEAC value of the paulownia extract was 0.821 ± 0.013 and the TEAC value of the positive control butylated hydroxytoluene (BHT) was 1.813 ± 0.022.
4. Total Antioxidant Activity Using FRAP Method
The FRAP method is one of the frequently used methods to check the antioxidant activity of a sample through its electron donating ability.
As a result of measuring the antioxidant power of paulownia extract by FRAP method, it showed 1.647 ± 0.018 mM FeSO4 eq./mg extract (Table 3).
The positive control group, BHT, showed a value of 1.363 ± 0.121 mM FeSO4 eq./mg extract. This indicates that the paulownia extract has a strong antioxidant power through a higher reducing power than the antioxidant BHT.
5. Cytotoxicity measurement
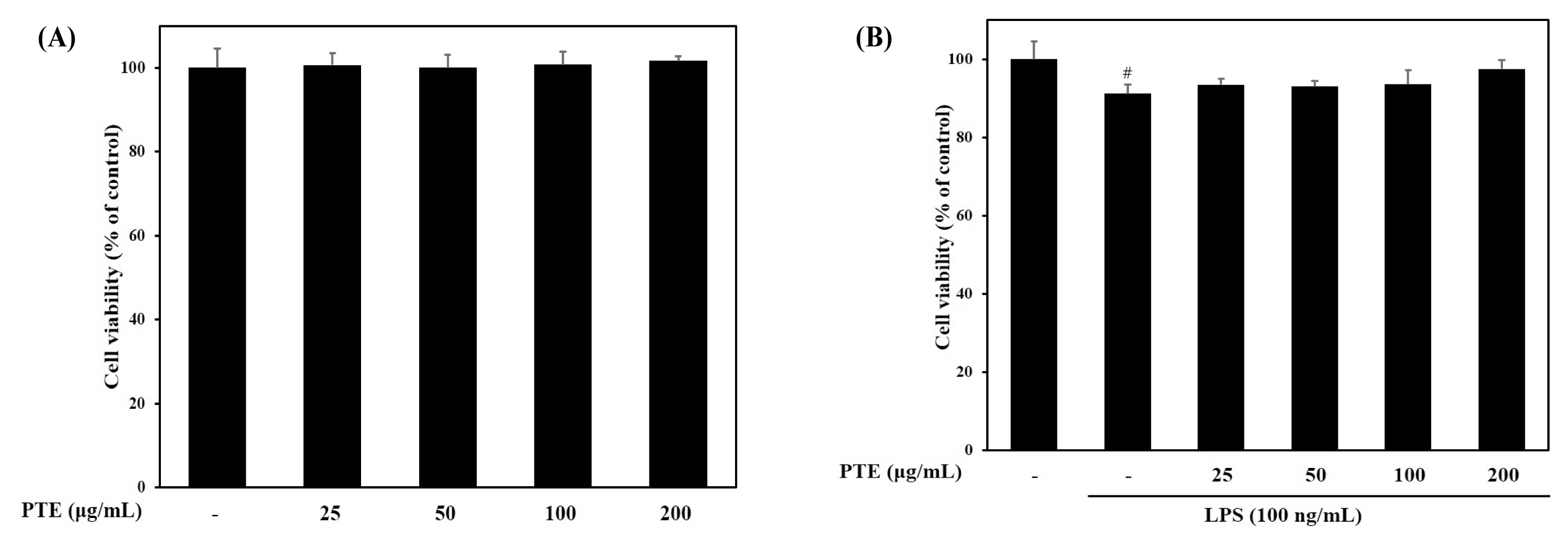
Cytotoxicity was examined to determine the effect of paulownia extract on RAW264.7 cells. Paulownia extract was treated at various concentrations (25, 50, 100, 200 μg/mL) and cell viability was measured.
Samples were pretreated an hour ago and cell viability after 24 hours treatment with LPS (100 ng / mL) was measured using MTT assay.
As a result, it was confirmed that there was no significant difference between the control and the cell viability after only treatment with the paulownia extract up to the concentration of 25–200 μg/mL (Fig. 1A).
In addition, LPS-treated group showed 92.19 ± 2.34% cell viability compared to the control group. However, the cell survival rate after pretreatment of the extract for one hour and LPS treatment did not show any significant difference. There was no significant difference from the control group not treated with LPS (Fig. 1B).
Therefore, Paulownia extract did not affect cell viability at concentrations of 200 μg/mL or less, and thus was determined to be non-toxic. Then the experiment was carried out at a concentration of 200 μg/mL or less.
6. Inhibitory Effect of Nitric Oxide (NO) Formation
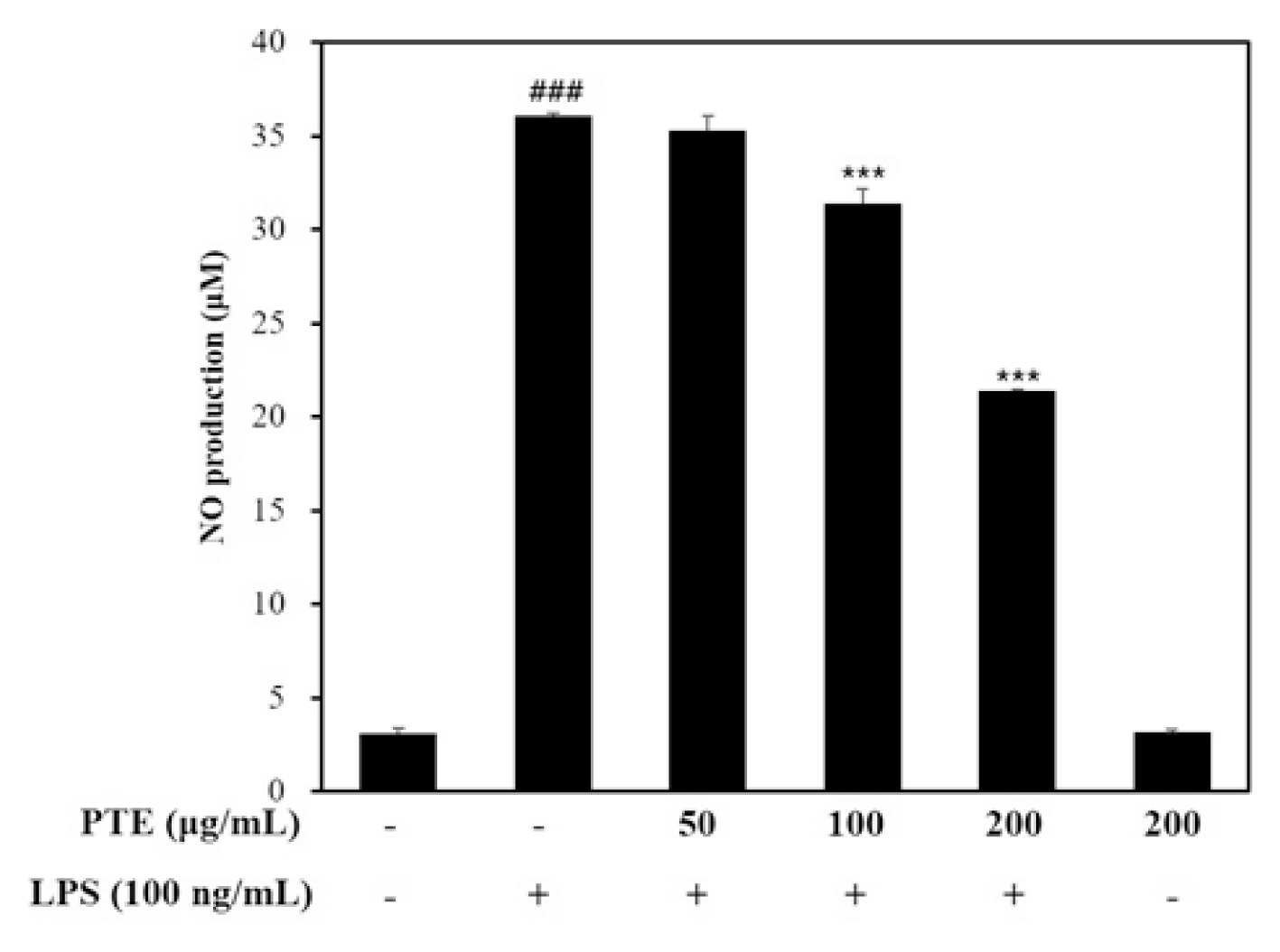
In this study, the effect of paulownia extract on NO production was examined. After 1 hour pretreatment of paulownia extract, NO production in RAW264.7 cells induced inflammatory reaction with LPS was measured using griess reagent.
It was measured at concentrations of extracts (50, 100, 200 μg / mL) that did not affect cell viability. When LPS was treated to induce inflammation, NO production was 36.08 ± 0.11 μ M. When treated with 200 μg/mL Paulownia extract, NO production was 21.379 ± 0.11 μM. This is about 40% NO production inhibitory effect (Fig. 2).
The results of this study indicate that paulownia extract inhibits NO production in a concentration-dependent manner which increased in LPS induced macrophages.
7. Inhibitory Effect of Pro-inflammatory Cytokines Production
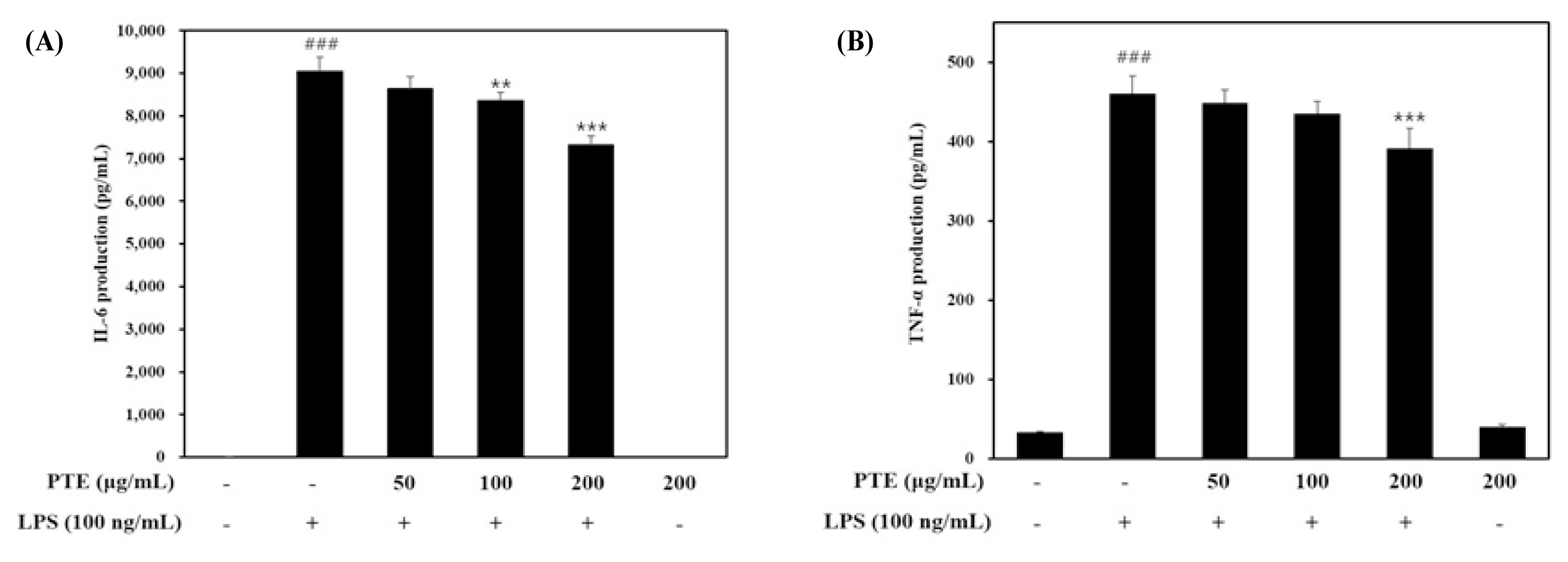
To investigate the effect of paulownia extract on cytokine secretion in LPS-induced inflammatory response RAW 264.7 cells, Paulownia extract was pretreated at various concentrations to measure changes in secretion levels of IL-6 and TNF-α using the ELISA kit.
As a result, it was confirmed that the secretion of cytokine increased by LPS was inhibited by paulownia extract (Fig. 3).
In the case of IL-6(Fig. 3A), the amount of secretion increased to 9037.77 ± 347.30 pg/mL by LPS was suppressed to 7311.85 ± 224.80 pg/mL when treated with 200 μg/mL Paulownia extract. This can be understood as an inhibitory effect of about 20%.
When inflammation was induced by LPS treatment, TNF-α showed a secretion amount of 460.18 ± 22.62 pg/mL. In comparison, in the group treated with 200 μg/mL Paulownia extract, the secretion was 391.13 ± 25.35 pg / mL, which was suppressed by 15% (Fig. 3B).
Through this, paulownia extract treatment seems to reduce of pro-inflammatory cytokines conentrations dependently.
8. Inhibitory Effect of Inflammation-related Protein Expression
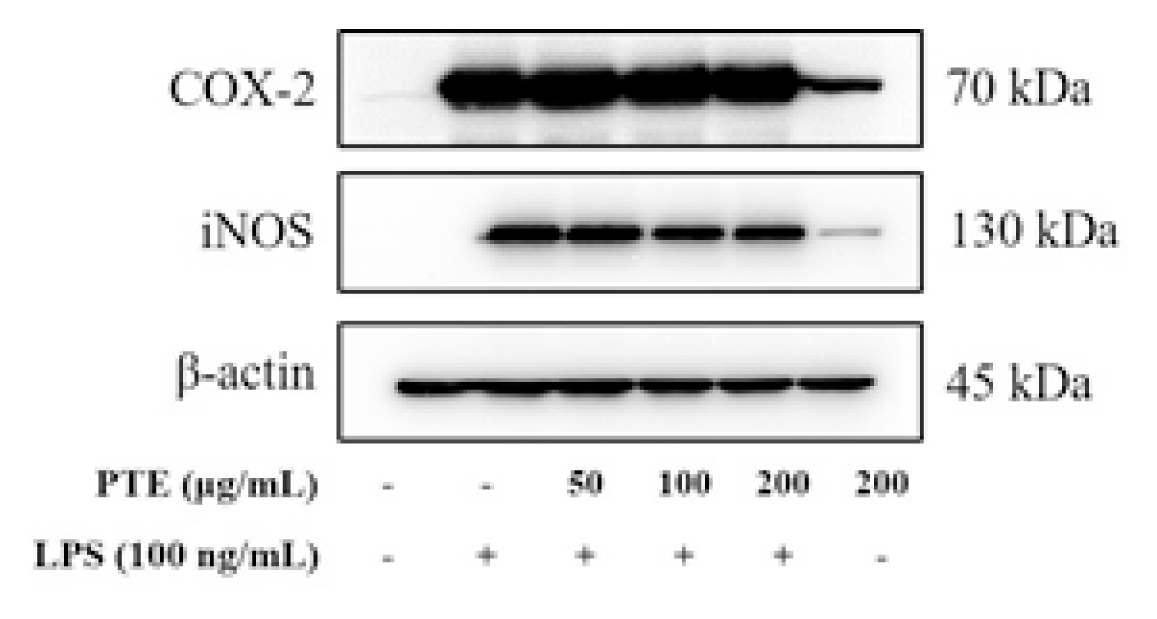
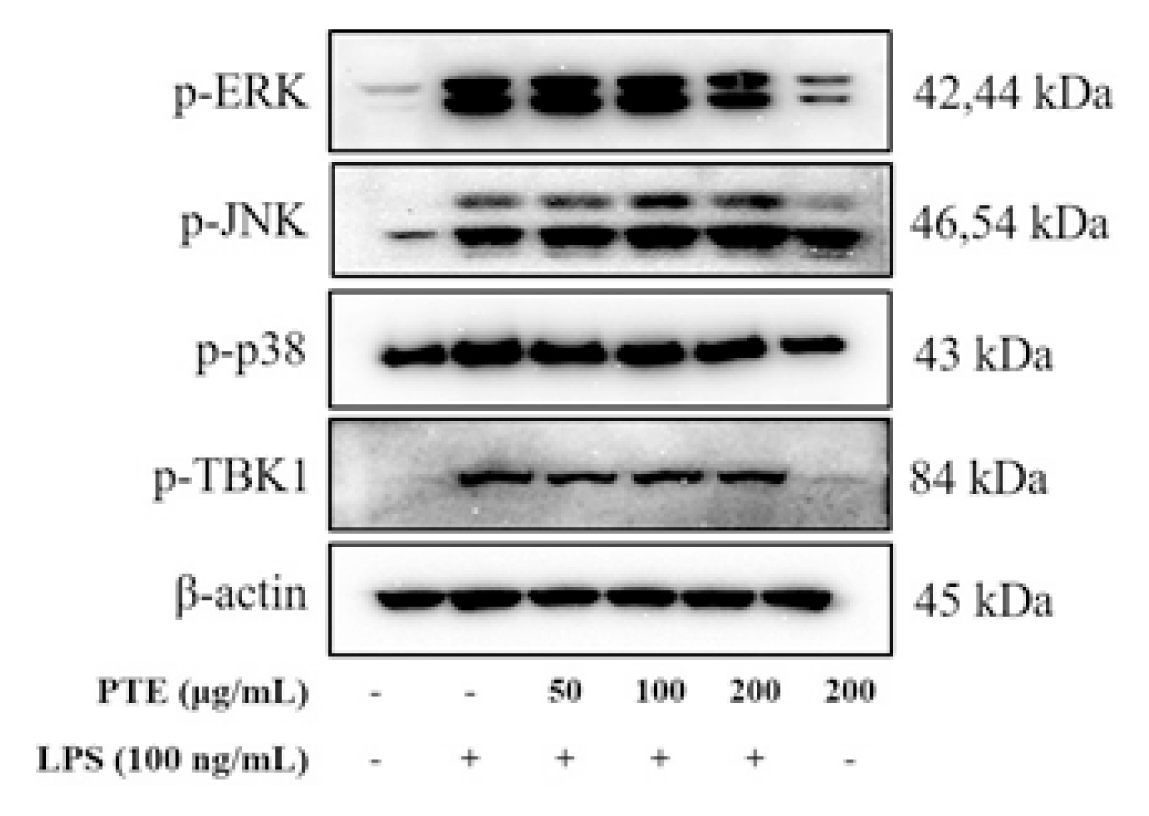
Western blot analysis was performed to investigate whether paulownia extracts influence the expression regulation of iNOS and COX-2. In the LPS-treated group, iNOS and COX-2 protein expression was significantly increased compared to the control group (Fig. 4).
In the group treated with paulownia extract, iNOS expression was reduced in a concentration -dependent manner and did not affect the expression inhibition of COX-2.
Signal transduction pathways of MAPKs (ERK, JNK, p38) play an important role in the activation of the inflammatory response. Particularly involved in inducing activation of transcription factors, including NF-κB and activator protein 1 (AP-1), increased secretion of inflammation-related mediators16,17).
The effect of paulownia extract on MAPKs expression was examined. LPS treatment significantly increased the expression of MAPK. The expression of p-ERK was markedly increased, but markedly decreased when 200 μg / mL of Paulownia extract was treated. However, p-p38 and p-JNK were not affected (Fig. 5). As a result of measuring the change in TBK1, a protein that affects MyD88 independent signaling, paulownia extract did not affect the increased p-TBK1 after LPS treatment (Fig. 5).
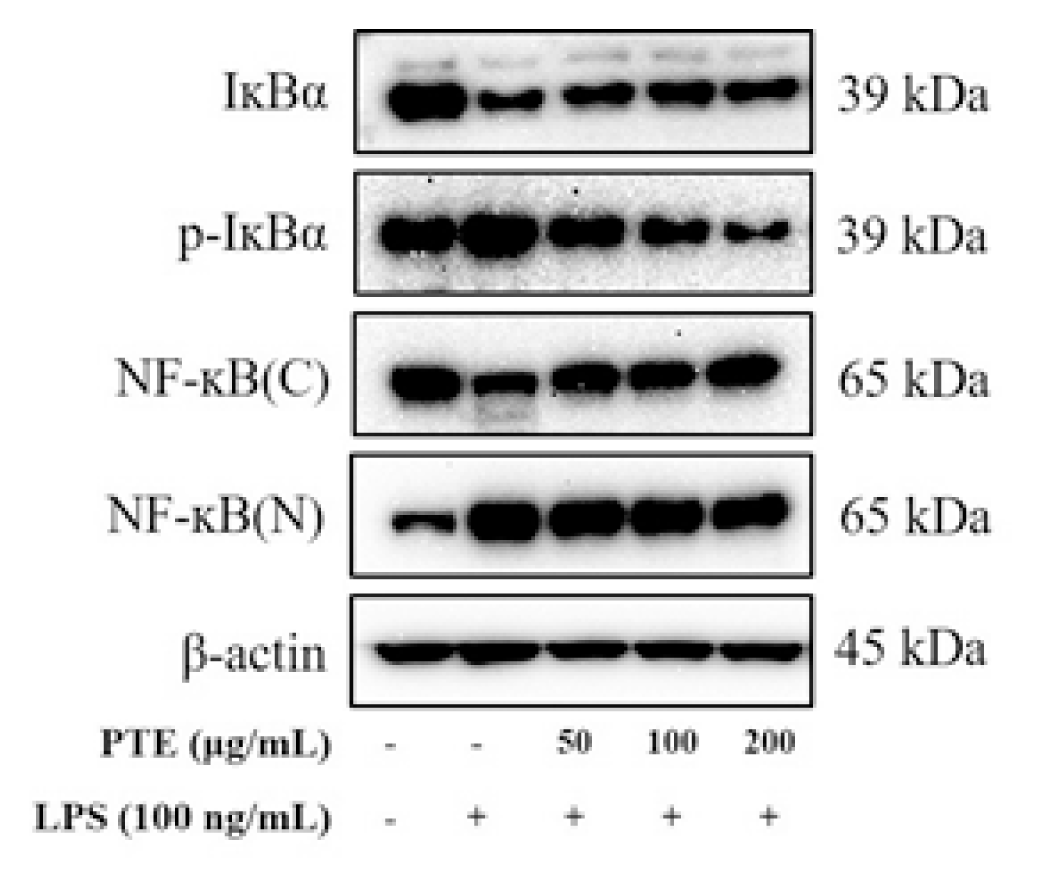
NF-κB is a transcriptional regulator that induces expression of inflammation-related mediators. When activated by LPS, NF-κB increases the expression of iNOS, COX-2, and pro-inflammatory cytokine18).
Normally, cells are present in the cytosol in the heterodimer form of NF-κB p65 and p50. When IκBα is phosphorylated by the inflammatory response, it moves to the nucleus and acts as a transcription factor19).
In this study, p-IκBα was significantly increased and the amount of IκBα was significantly decreased in the LPS-induced group (Fig. 6).
However, when the paulownia extract was treated, it was confirmed that the phosphorylation of IκBα was inhibited. In addition, the expression of NF-κB p65 protein in the nuclear fraction showed that in the LPS-treated group, the intranuclear migration of NF-κB was significantly increased in comparison with the control group and decreased in the paulownia extract-treated group (Fig. 6).
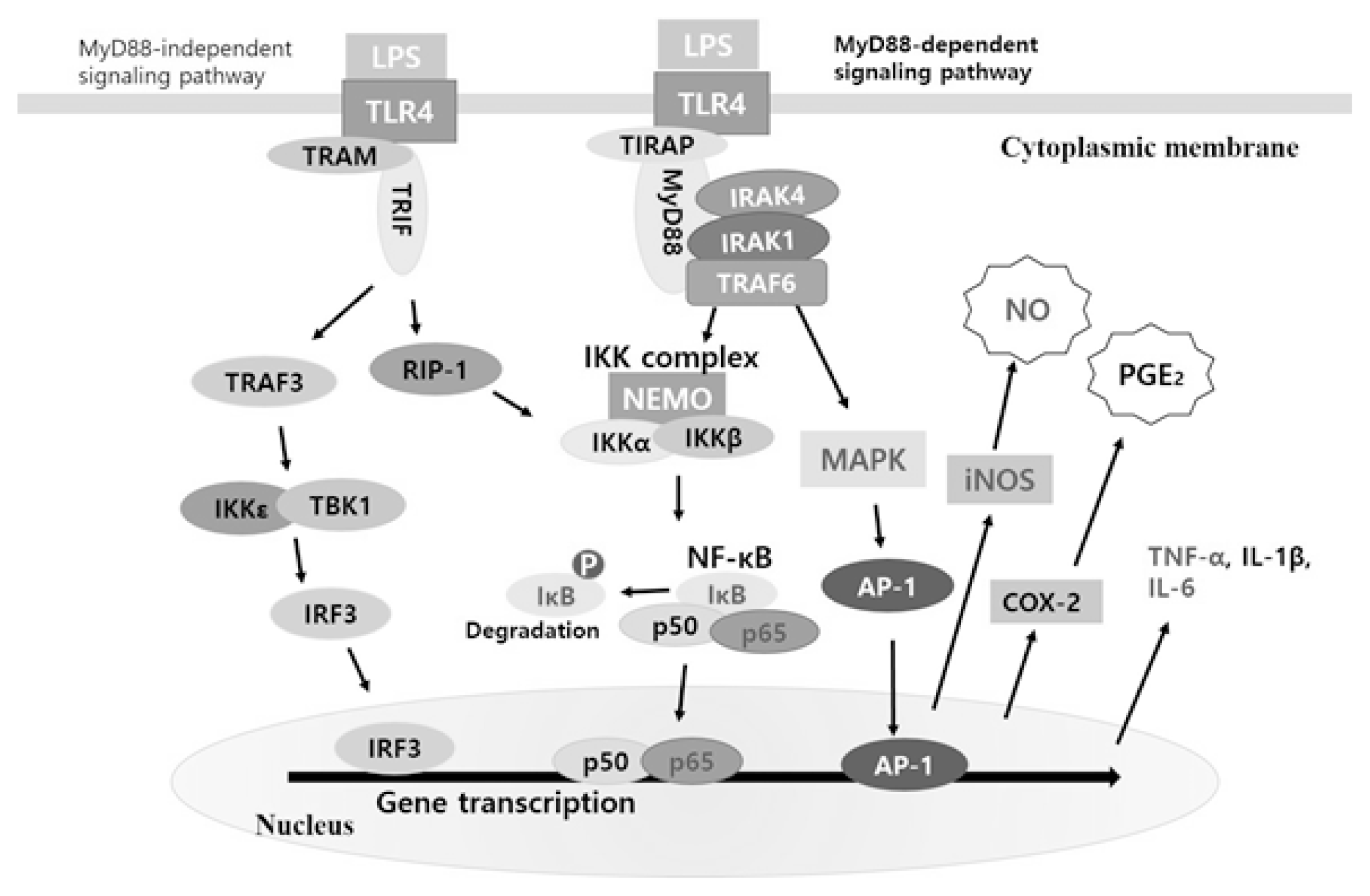
As a result, it was confirmed that the paulownia extract has anti-inflammatory effect by inhibiting the phosphorylation of IκBα in LPS-activated macrophages, inactivating signaling pathways that inhibit the migration of NF-κB p65 in the nucleus. Therefore, based on the results of this study, the MyD88 dependent pathway schema related to the anti-inflammatory efficacy of paulownia extract is shown in Fig. 7.
9. Summary
The purpose of this study was to determine the antioxidant activity of paulownia extract and to determine the total polyphenol and flavonoid content in paulownia extract. The total polyphenol content was 148.98 ± 1.84 mg GAE/g extract and the total flavonoid content was 115.33 ± 4.16 mg CE/g extract.
As a result of measuring DPPH radical scavenging ability using ESR, the IC50 value of 50% radical scavenging concentration was 0.025 mg/mL, indicating high DPPH radical scavenging activity.
Radical scavenging activity using ABTS and total antioxidant activity using FRAP method also showed that Paulownia extract had high antioxidant effect. RAW264.7 cells induced inflammation with LPS were treated with paulownia extract at non-toxic concentrations to determine NO, an inflammatory mediator. As a result, it was confirmed that the amount of NO produced was reduced in a concentration -dependent manner.
In addition, the production of pro-inflammatory cytokine (IL-6, TNF-α) was inhibited, iNOS expression was also reduced.
Paulownia extract also affected the phosphorylation of ERK through the MyD88 dependent signal transduction pathway, a major pathway of the inflammatory response. In addition, the inhibition of phosphorylation of IκB α inhibited nuclear migration of NF-κB, which showed an anti-inflammatory effect.
The results of this study can provide basic information on anti-inflammatory materials derived from Korean native plants. Further studies on the separation and purification of active compounds with anti-inflammatory effects are expected.