Introduction
Tuberous sclerosis complex (TSC) is one of the most common types of neurocutaneous syndrome. Its major features include tumor-like lesions called hamartomas showing up in brains, hearts, kidneys or on skin.1) The most common neurologic disorder that occurs in patients with TSC is seizure. Approximately 80–90 % of TSC patients undergo seizures; in most cases, the onset of the first seizure occurs before the age of one year and they often accompany infantile spasms (IS). Early onset seizures and drug-resistant seizures like IS are known to leave more severe neuro-developmental and cognitive impairments.2)
Yukmijihwang-tang is traditionally widely used as a prescription for mental and physical development delay in East Asia. In preclinical studies, it was found to increase neurogenesis and spatial memory functions and have neuro-protective effects.3–4) Cheonma and Jogudeung, two other herbal ingredients, have anticonvulsant effects such that they are now receiving attention as less toxic alternatives of standard antiepileptic drugs (AEDs).5–6) At Dong Seo Yung Hap (DSYH) hospital, by the process of the distinct diagnostic method in Korean traditional medicine, modified Yukmijihwang-tang (YMJ)-Cheonma and Jogudeung added to Yukmijihwang-tang - is used as one of the treatments for epilepsy.
This case report was written to detail on the YMJ treatment on an infant with TSC and intractable epilepsy that resulted in a significantly positive seizure control and developmental improvement.
Case
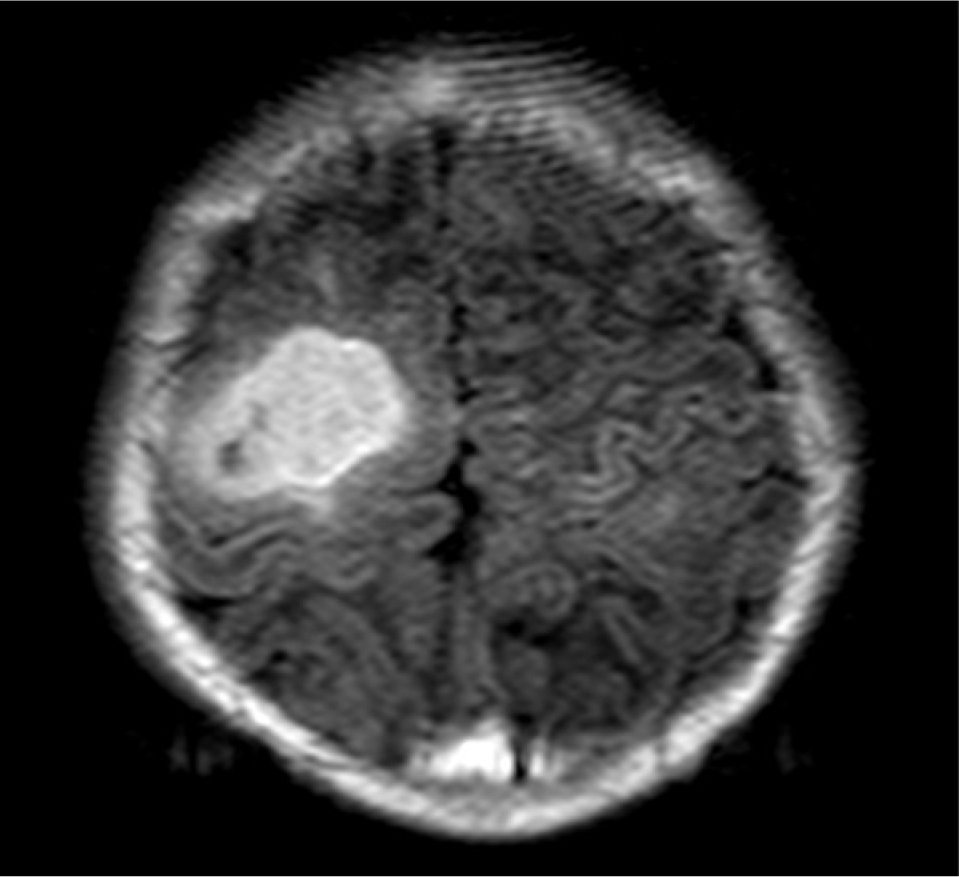
Born a boy after a gestational period of 40 weeks by normal spontaneous vaginal delivery weighing 2.85 kg, the subject was diagnosed with cardiac rhabdomyoma before birth and experienced his first seizure at his birth. The electroencephalogram (EEG) test detected nearly continuous epileptiform discharge in the right central area of his brain and diagnosed the subject with partial seizure two days after his birth. Accordingly, he began to take levetiracetam. The MRI test detected various sizes of tubers in the cortex and subependymal area, with the biggest lesion in his right frontal lobe at 2.5 cm (Figure 1). More than three hypomelanotic macules were observed throughout his body, and a genetic analysis showed deletion frameshift mutation in TSC2 gene which definitively confirmed the diagnosis of tuberous sclerosis. All of these tests and medications are conducted at the Department of Pediatrics, A Medical Center, University of U College of Medicine, Seoul.
A month after birth, he experienced seizures that were indicative of epileptic spasms as well as partial seizure. In the follow-up EEG, modified hypsarrythmia was confirmed and vigabatrin was added to his prescription with the IS diagnosis. However, epileptic spasms continued and developmental regression signs such as decreased eye-contact, cooing and smiling began to show.
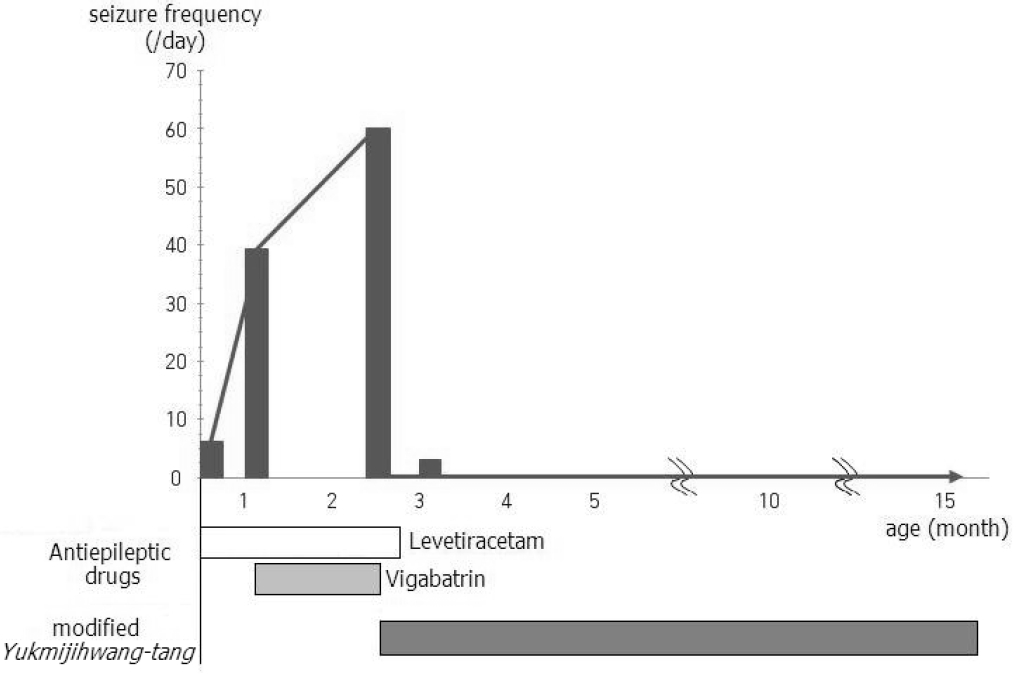
At 2.5 months after birth (January 31th, 2014) when the subject first visited DSYH hospital he was taking 50 mg of levetiracetam twice daily and 500 mg of vigabatrin twice a day. The subject was experiencing around 6–7 clusters with approximately 10 spasms per cluster, with a total of 60–70 epileptic spasms per day, and displaying developmental regression and delay. He showed levels of under one-month old in all five areas of the Korean Infant and Child Developmental Test (KICDT), which was developed to evaluate the development status of infant and child in terms of developmental age (DA) and developmental quotient (DQ).7) He began to take YMJ and stopped using vigabatrin provisionally.
YMJ’s daily dosage consists of the following: 4g of Rehmanniae Radix (地黃), 3g of Dioscorae Radix (山藥), 3g of Corni Fructus (山茱萸), 2g of Alimatis Rhizoma (澤瀉), 2g of Moutan Cortex Radicis (牧丹 皮), 2g of Hoelen (茯苓), 1.5g of Gastrodia elata (天 麻), and 1.5g of Uncaria sinensis (釣鉤藤). These herbal medicinal ingredients were boiled in approximately 100 cc of water at 102–103 °C for approximately 2 hours such that there was about 30 cc of liquid left. The solution was packaged by 10 cc and the subject was ordered to take it 3 times a day, 30 minutes after each meal.
Despite the discontinuation of vigabatrin usage, seizure frequency began to decrease with the use of YMJ; at one week of the YMJ treatment, the subject achieved cessation of spasms (Figure 2). Furthermore, when his movements increased and his eye-contact and neck-control began to improve, the infant’s guardians decided to also stop the use of levetiracetam provisionally. Two weeks after withdrawing from levetiracetam the subject showed approximately 3–5 partial seizures per day over a week, each one lasting around 30 seconds. However, he continued to show positive developmental improvement with eye-tracking, responding to sound or his name and thus his progress was observed without using AEDs. He reclaimed the seizure-free state after this period.
At 4 months old (1.5 month of YMJ treatment), when the child underwent the follow-up EEG at the general hospital at which he was first diagnosed with TSC, the test rendered resolution of modified hypsarrythmia. Moreover, the epilepticform discharge that was consistently present at the right central area of his brain was rarely observed this time.
At 5 months old, the child began rehabilitation treatment because the results of the KICDT test performed at DSYH hospital showed impaired fine motor skills of his left arm and hand. At 8 months old, he was diagnosed with left hemiplegia at the general hospital, which was assumed to be caused by TSC but by 14 months, he showed progress in his motor, language and cognitive-adaptive abilities, comparatively more in the latter two than the first (Table 1).
Last follow-up was conducted on May 15th, 2015 (18 months old). Until that time, the infant was continuing the YMJ treatment and rehabilitation for hemiplegia. He has not experienced side effects or adverse events during the past 15 months of treatment, and the trimonthly tests conducted on aspartate transaminase (AST), alanine transaminase (ALT), alkaline phospatase (ALP), gamma glutamyl tranapeptidase (GGT), total protein, albumin, total bilirubin, blood urea nitrogen (BUN), and creatinine levels all rendered results in the normal range.
Discussion
Infants with TSC experiencing IS are reported to show approximately 85–100 % probability of mental retardation.8–10) In particular, patients with TSC2 gene mutation experience more tubers, earlier seizure onset, and have higher probability of IS and intractable seizures - which could all lead to more profound intellectual disability - than those with TSC1 gene mutation.11) In general, infants with TSC displaying IS are typically treated first with vigabatrin12), which shows approximately 50–90 % clinical response rate.13)
The subject in this study was found to have TSC2 gene mutation, began experiencing neonate seizure and progressed to IS, and was deemed to have a type of refractory epilepsy that does not respond to vigabatrin and levetiracetam. The fact that he achieved the seizure-free state without side effects or that he became “normal” by the EEG test after discontinuing the use of AEDs and only taking YMJ shows that YMJ has seizure control effects.
The subject also showed developmental progress only with the use of YMJ; this progress was far more significant in language and cognitive-adaptive abilities. Such result is likely due to the patient having a greater motor ability impairment and/or YMJ having more influence in the areas involving intellectual abilities rather than motor abilities.
Intellectual impairments that show up in TSC patients are generally attributed to the underlying pathology of TSC itself or to seizure. Most research up to this point has been focused on seizure control; however, the proportion of those whose cognitive impairment is due to seizure is reported to be below 50 %.2) Hence, research on mammalian target of rapamycin (mTOR) inhibitors that directly act on TSC’s pathophysiological mechanisms are rendering some more productive results, though with limitations.14) Considering that developmental regression and delay were observed in this study’s subject while IS was in progress, and that his developmental improvement began after he became seizure-free with the YMJ treatment, seizure control seems to have at least a limited amount of influence on development outcomes. However, seeing as Yukmijihwang-tang, the largest part of YMJ’s composition, has traditionally been used as a treatment for infant developmental impairment rather than for epilepsy, YMJ may have been more affective in developmental improvement from directly affecting the underlying pathology of TSC. More clinical and preclinical studies on such matters are needed in the near future.