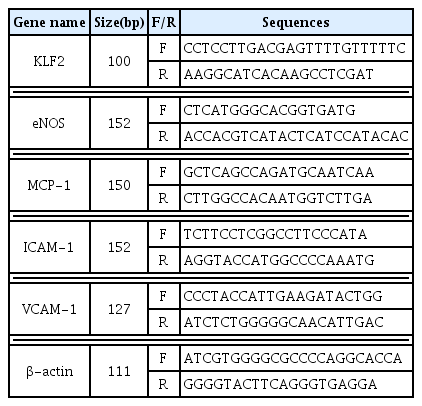
Anti-inflammatory Effect of Cornus Officinalis fruit extract and Cornus Officinalis Fruit Cheonghyeol Plus in Human Umbilical Vein Endothelial Cell
Article information
Abstract
Objectives
The purpose of this study was to investigate the anti-inflammatory effect of Cornus Officinalis fruit extract(CE) and Cornus Officinalis Fruit Cheonghyeol Plus(CCP) in Human Umbilical Vein Endothelial Cell.
Methods
We measured cell viability of CE, CCP and treated HUVEC with TNF-α. We measured the mRNA expression levels of KLF2, eNOS, MCP-1, ICAM-1, VCAM-1, the protein expression levels of KLF2, eNOS, MCP-1, ICAM-1, VCAM-1, and the protein phosphorylation level of ERK, JNK, p38 and the biomarker expression levels of MCP-1, ICAM-1, VCAM-1.
Results
1.CE incresed the mRNA, protein expression levels of KLF2, eNOS at concentrations of 100μg/ml compared to the control group. CE decresed the mRNA, protein and biomarker expression levels of MCP-1,ICAM-1,VCAM-1 at concentrations of 100μg/ml compared to the control group. CE decresed the protein phosphorylation level of p38 at concentrations of 100μg/ml compared to the control group. 2. CCP incresed the mRNA, protein expression levels of KLF2, eNOS at concentrations of 100μg/ml or more compared to the control group. CCP decresed the mRNA, protein and biomarker expression levels of MCP-1, ICAM-1, VCAM-1 at concentrations of 100μg/ml or more compared to the control group. CCP decresed the protein phosphorylation level of ERK at concentrations of 100μg/ml or more, p38 at concentrations of 200μg/ml or more, and JNK at concentrations of 400μg/ml compared to the control group.
Conclusions
These results present that CE and CCP has anti-inflammatory effect in HUVEC. So, it could help treat or prevent inflammation in vein caused by dyslipidemia and contribute prevention of cardiovascular and cerebrovascular cerebrovascular diseases.
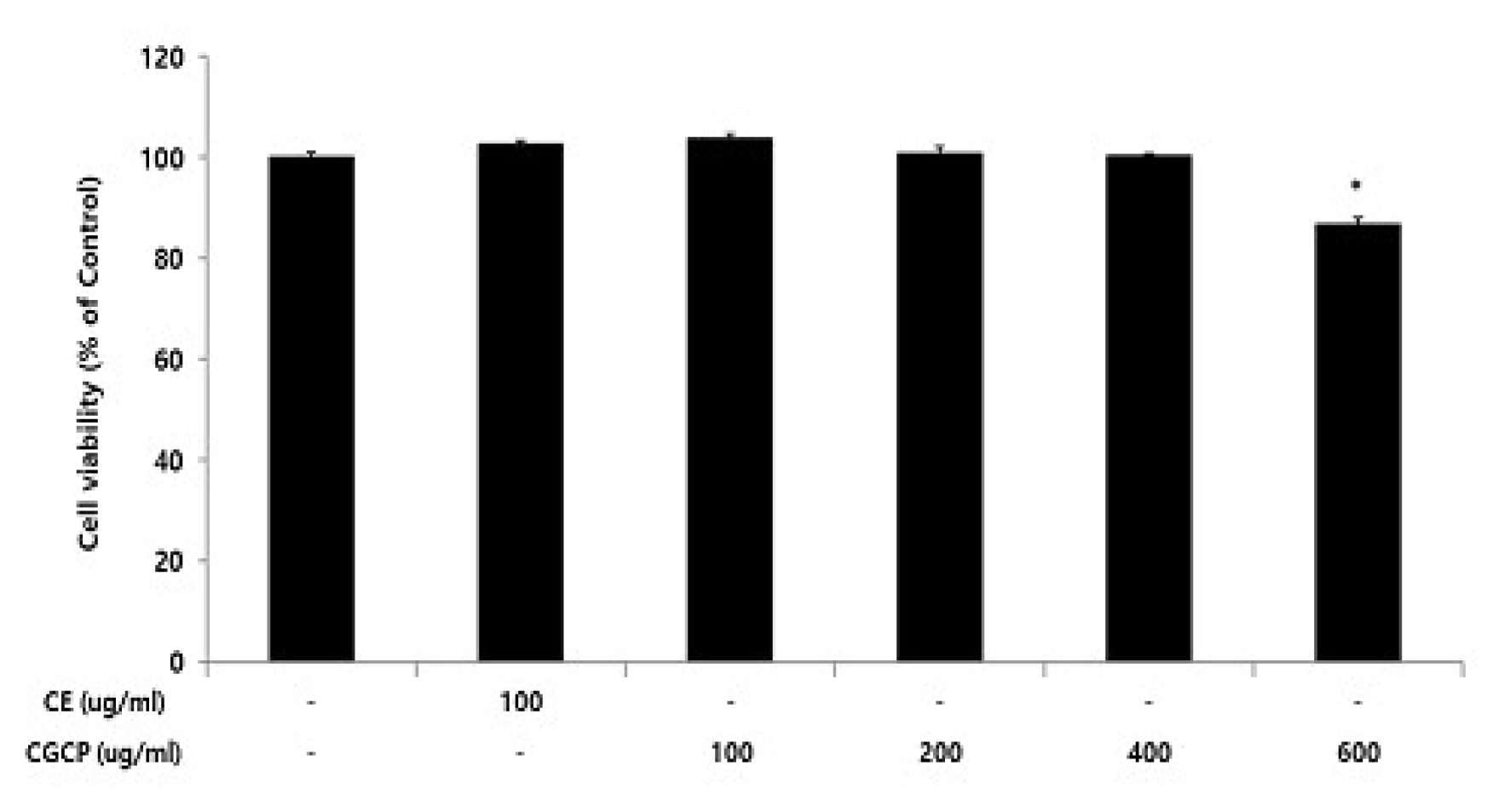
Cell viability of CE or CCP in HUVEC
Cell viability were calculated as percentage relative to the control and the result were given by the mean±standard error of mean from three independent experiments(Significance of results, *; p<0.05 compared to control).

Effect of CE or CCP on KLF2(A), eNOS(B), MCP-1(C), ICAM-1(D), VCAM-1(E) mRNA expression level in HUVEC
HUVEC were treated CE(100 ug/ml or CCP(100, 200, and 400 ug/ml), with TNF-α 10 ng/ml for 12hour. The mRNA expression level were measured using a real-time PCR(qPCR). The result were given by the mean±standard error of mean from three independent experiments(Significance of results, *; p<0.05, **; p<0.01, ***; p<0.001 compared to control, +++; p<0.001 compared to normal).
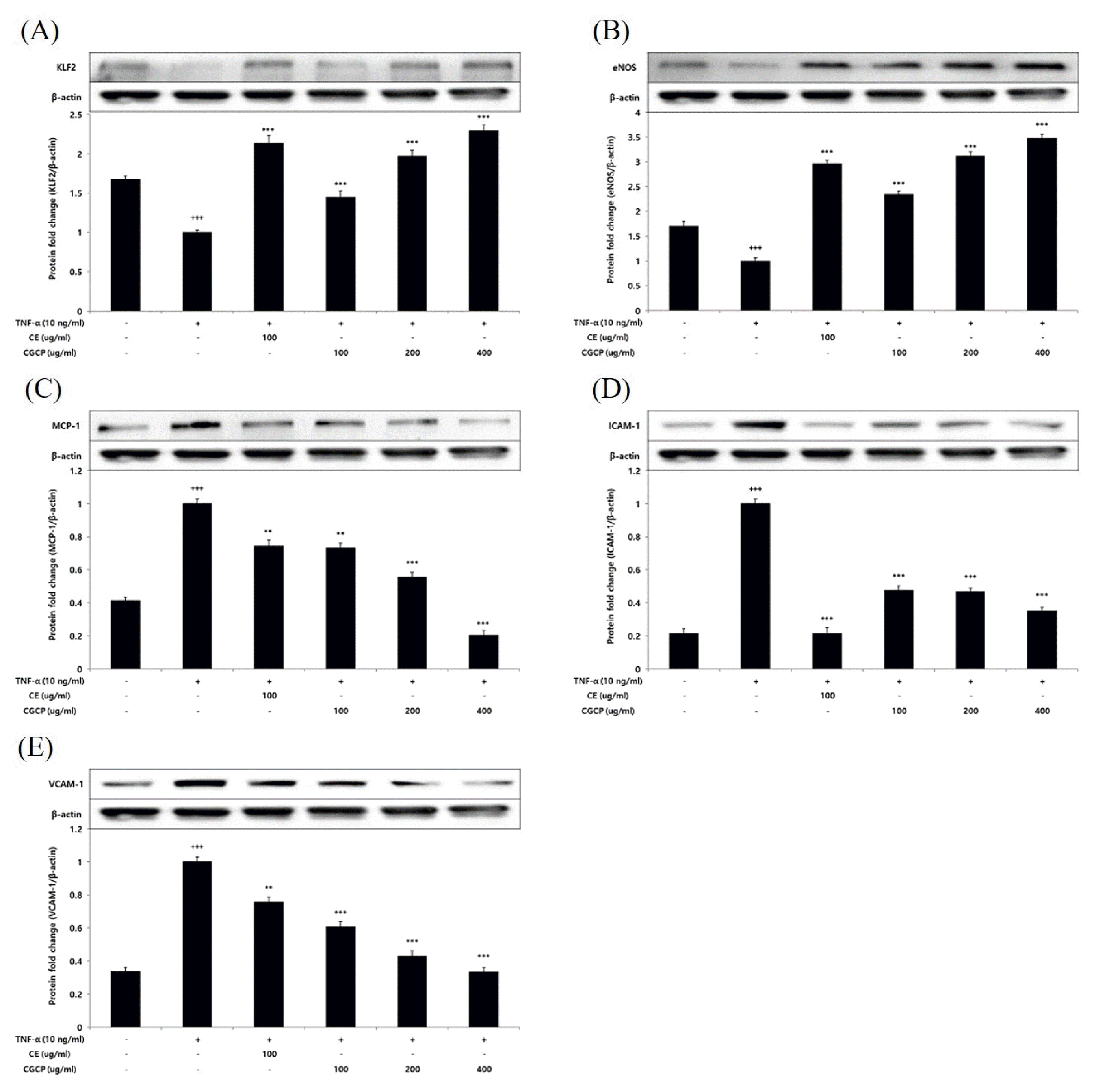
Effect of CE or CCP on KLF2(A), eNOS(B), MCP-1(C), ICAM-1(D), VCAM-1(E) protein expression level in HUVEC
HUVEC were treated CE(100ug/ml or CCP(100, 200, and 400ug/ml), with TNF-α 10 ng/12 hour. The protein expression level were measured by a western blot. The result were presented by the mean±standard error of mean from three independent experiments(Significance of results, *; p<0.05, **; p<0.01, ***; p<0.001 compared to control, +++; p<0.001 compared to normal).
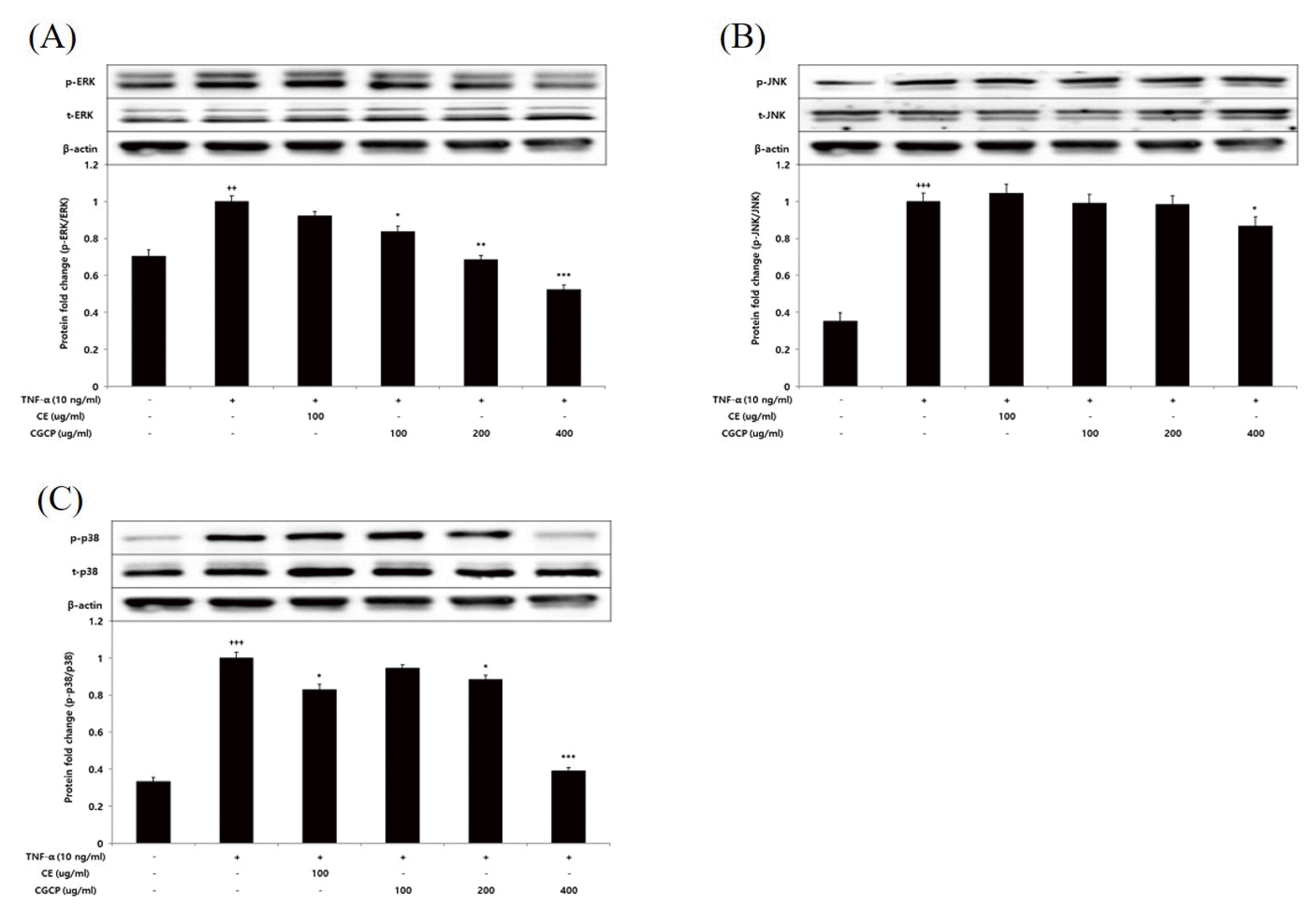
Effect of CE or CCP on ERK(A), JNK(B), p38(C) protein phosphorylation level in HUVEC
HUVEC were treated CE(100ug/ml or CCP(100, 200, and 400ug/ml), with TNF-α 10 ng/12 hour. The protein phosphorylation level were measured by a western blot. The result were presented by the mean±standard error of mean from three independent experiments(Significance of results, *; p<0.05, **; p<0.01, ***; p<0.001 compared to control, +++; p<0.001 compared to normal).
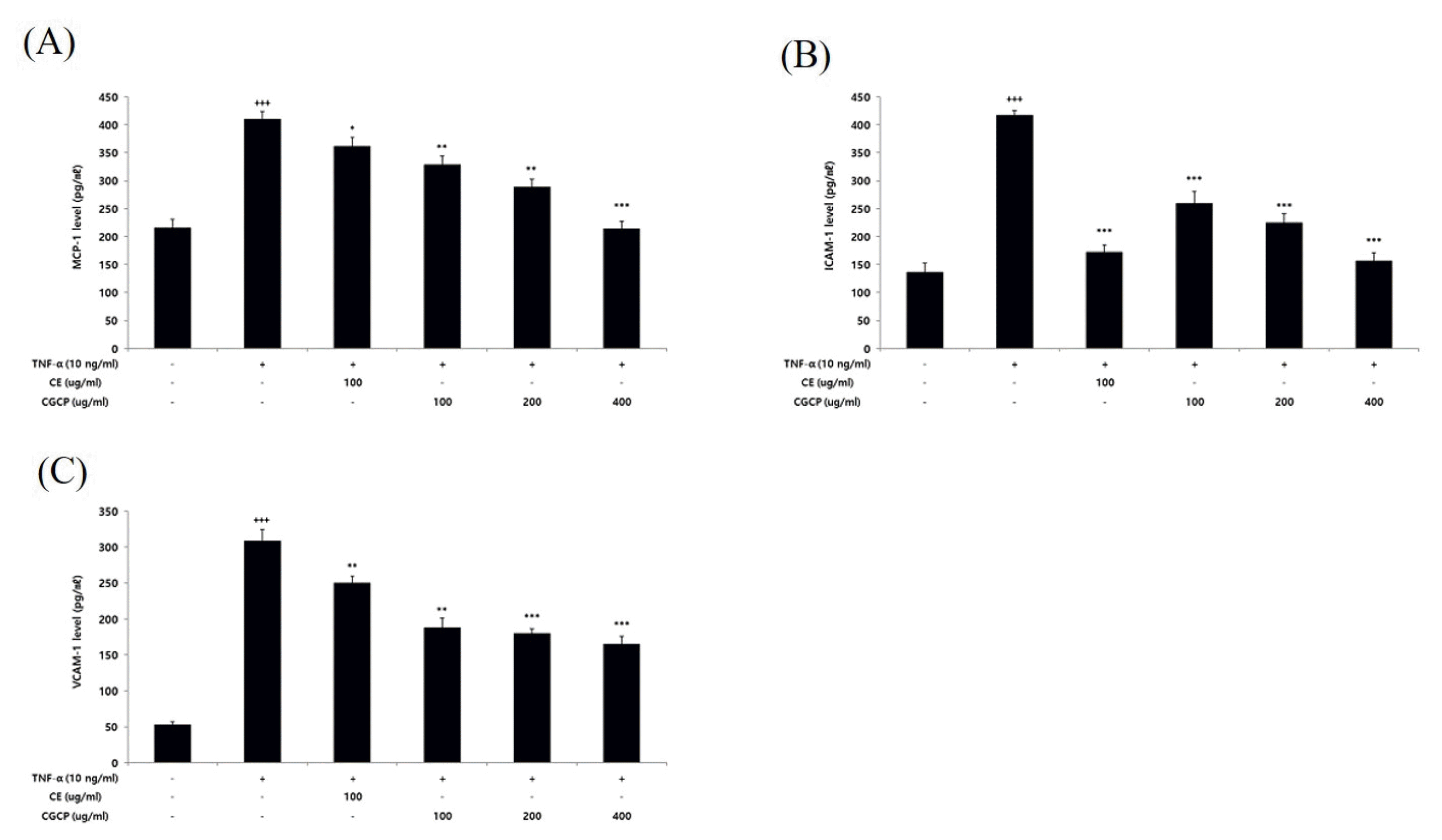
Effect of CE or CCP on MCP-1(A), ICAM-1(B), VCAM-1(C) level in HUVEC
HUVEC were treated CE(100ug/ml or CCP(100, 200, and 400ug/ml), with TNF-α 10 ng/12hour. MCP-1 level was measured by ELISA kit. The result were presented by the mean±standard error of mean from three independent experiments(Significance of results, *; p<0.05, **; p<0.01, ***; p<0.001 compared to control, +++; p<0.001 compared to normal).
CE : Cornus Officinalis fruit extract
CCP : Cornus Officinalis Fruit Cheonghyeol Plus
HUVEC : Human Umbilical Vein Endothelial Cell
KLF : Kruppel-like factor 2
eNOS : Endothelial nitric oxide synthase
MCP-1 : Monocyte chemoattractant protein-1
ICAM : Intercellular Adhesion Molecule -1
VCAM : Vascular Cell Adhesion Molecule -1
TNF : Tumor necrosis factor-α
ERK : Extracellular signal-regulated kinase
JNK : Jun N-terminal kinase