References
1. Lagman RL, Davis MP, LeGrand SB, Walsh D. Common Symptoms in Advanced Cancer. Surg Clin North Am 2005;Apr. 85(2):237–55.
2. Mantovani G, Macci? A, Massa E, Madeddu C. Managing Cancer-Related Anorexia/Cachexia. Drugs 2001;61(4):499–514.
3. Wie G-A, Cho Y-A, Kim S-Y, Kim S-M, Bae J-M, Joung H. Prevalence and risk factors of malnutrition among cancer patients according to tumor location and stage in the National Cancer Center in Korea. Nutr Burbank Los Angel Cty Calif 2010;Mar. 26(3):263–8.
4. Brierley DI, Harman JR, Giallourou N, Leishman E, Roashan AE, Mellows BAD, et al. Chemotherapy-induced cachexia dysregulates hypothalamic and systemic lipoamines and is attenuated by cannabigerol. J Cachexia Sarcopenia Muscle 2019;Apr. 29. jcsm.12426.
5. Lis CG, Gupta D, Grutsch JF. Can anorexia predict patient satisfaction with quality of life in advanced cancer? Support Care Cancer Off J Multinatl Assoc Support Care Cancer 2009;Feb. 17(2):129–35.
6. Nelson KA, Walsh D, Sheehan FA. The cancer anorexia-cachexia syndrome. J Clin Oncol Off J Am Soc Clin Oncol 1994;Jan. 12(1):213–25.
7. Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, et al. ESPEN guidelines on nutrition in cancer patients. Clin Nutr Edinb Scotl 2017;36(1):11–48.
8. Arends J, Baracos V, Bertz H, Bozzetti F, Calder PC, Deutz NEP, et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin Nutr Edinb Scotl 2017;36(5):1187–96.
9. White JD. Complementary and alternative medicine research: a National Cancer Institute perspective. Semin Oncol 2002;Dec. 29(6):546–51.
10. Mao JJ, Palmer CS, Healy KE, Desai K, Amsterdam J. Complementary and alternative medicine use among cancer survivors: a population-based study. J Cancer Surviv 2011;Mar. 5(1):8–17.
11. Lau CHY, Wu X, Chung VCH, Liu X, Hui EP, Cramer H, et al. Acupuncture and Related Therapies for Symptom Management in Palliative Cancer Care: Systematic Review and Meta-Analysis. Medicine (Baltimore) 2016;Mar. 95(9):e2901.
12. Huang Z, Qin Z, Yao Q, Wang Y, Liu Z. Moxibustion for Chemotherapy-Induced Nausea and Vomiting: A Systematic Review and Meta-Analysis. Evid-Based Complement Altern Med ECAM 2017;20179854893.
13. Lee J-H, Bae K, Yoo H-S. The Effect of Traditional Oriental Herbal Medicine for Anorexia in Cancer Patients: A Systematic Review. J Korean Med 2017;Mar. 31. 38(1):8–20.
14. Han S, Jang B-H, Hwang D-S, Suh HS. Experiences of Treatment-Related Side Effects and Supportive Care with Korean Medicine in Women with Breast Cancer - A Focus Group Study. J Orient Obstet & Gynecol 2017;30(1):85–94.
15. Choi Y-J, Lee J-S, Cho S-H. Use of Korean Medicine among Cancer Patients. J Korean Orient Med 2012;Sep. 33(3):46–59.
16. Kroenke K, Monahan PO, Kean J. Pragmatic Characteristics of Patient-Reported Outcome Measures are Important for Use in Clinical Practice. J Clin Epidemiol 2015;Sep. 68(9):1085–92.
17. Shih Y-L, Hsu Y-C, Chiu W-H, Chao C-SC. Estimating Life Expectancy From Chinese Medicine Could Improve End-of-Life Care in Terminally Ill Cancer Patients. Holist Nurs Pract 2018;Oct. 32(5):247–52.
18. Meropol NJ, Egleston BL, Buzaglo JS, Benson AB, Cegala DJ, Diefenbach MA, et al. Cancer Patient Preferences for Quality and Length of Life. Cancer 2008;Dec. 15. 113(12):3459–66.
19. Cho J-H, Son C-G, Lee Y-W, Yoo H-S, Sung J-H, Kim S-Y, et al. Current Status of Studies on Cancer-Related Quality of Life Instruments. J Korean Orient Intern Med 2006;27(1):84–91.
20. Lee J-A, Lee S-H, Park J-H, Park J-H, Kim S-G, Seo J-H. Analysis of the Factors Related to the Needs of Patients with Cancer. J Prev Med Pub Health 2010;43(3):222.
21. Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;Dec. 5(6):649–56.
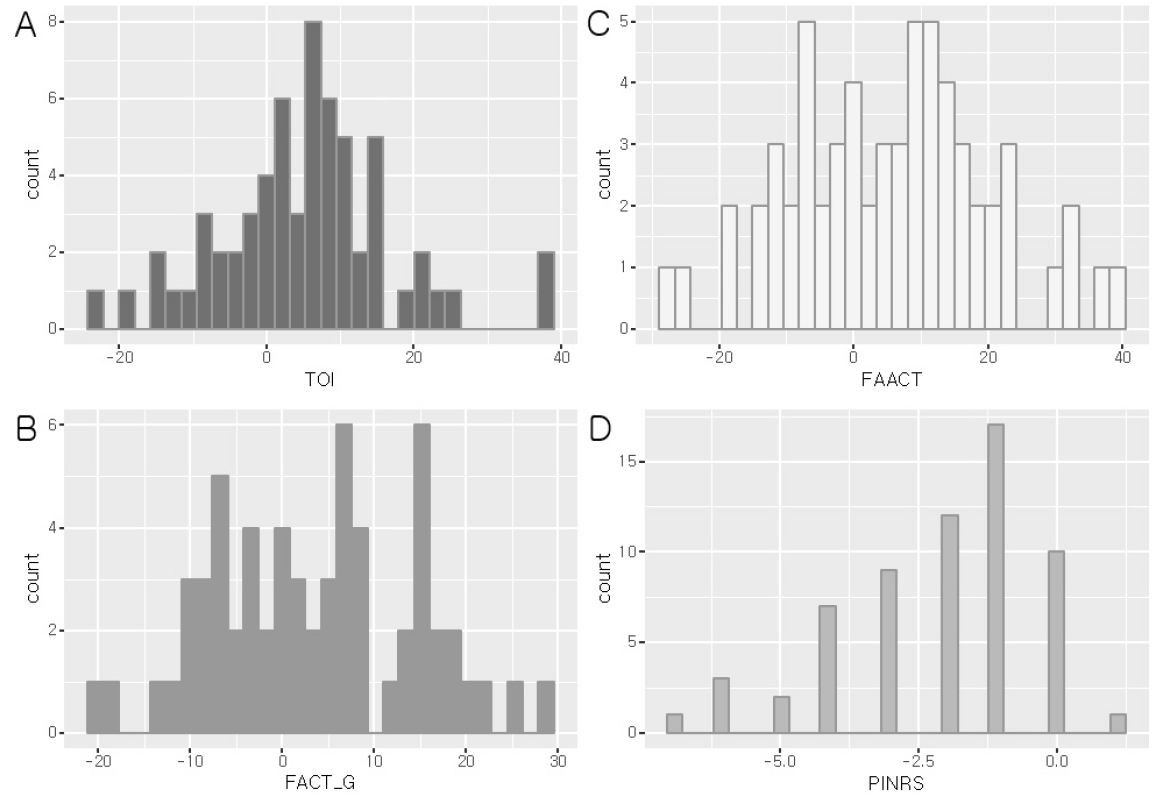
22. Ribaudo JM, Cella D, Hahn EA, Lloyd SR, Tchekmedyian NS, Roenn JV, et al. Re-validation and Shortening of the Functional Assessmentof Anorexia/Cachexia Therapy (FAACT) Questionnaire. Qual Life Res 2001;9(10):1137–46.
23. Cella DF, Tulsky DS, Gray G, Sarafian B, Linn E, Bonomi A, et al. The Functional Assessment of Cancer Therapy scale: development and validation of the general measure. J Clin Oncol 1993;Mar. 11(3):570–9.
24. Cella DF, Bonomi AE, Lloyd SR, Tulsky DS, Kaplan E, Bonomi P. Reliability and validity of the functional assessment of cancer therapy-lung (FACT-L) quality of life instrument. Lung Cancer 1995;Jun. 12(3):199–220.
25. Farrar JT, Young JP, LaMoreaux L, Werth JL, Poole MR. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain 2001;Nov. 94(2):149–58.
26. Guy W. ECDEU assessment manual for psychopharmacology. Rev. 1976 Rockville, Md: U.S. Dept. of Health, Education, and Welfare, Public Health Service, Alcohol, Drug Abuse, and Mental Health Administration, National Institute of Mental Health, Psychopharmacology Research Branch, Division of Extramural Research Programs; 1976. p. 603.
27. Webster K, Cella D, Yost K. The Functional Assessment of Chronic Illness Therapy (FACIT) Measurement System: properties, applications, and interpretation. Health Qual Life Outcomes 2003;16(1):7.
28. Tranmer JE, Heyland D, Dudgeon D, Groll D, Squires-Graham M, Coulson K. Measuring the Symptom Experience of Seriously Ill Cancer and Noncancer Hospitalized Patients Near the End of Life with the Memorial Symptom Assessment Scale. J Pain Symptom Manage 2003;May. 25(5):420–9.
29. Tisdale MJ. Mechanisms of Cancer Cachexia. Physiol Rev 2009;Apr. 89(2):381–410.
30. Ruiz Garcia V, López-Briz E, Carbonell Sanchis R, Gonzalvez Perales JL, Bort-Martí S. Megestrol acetate for treatment of anorexia-cachexia syndrome. Cochrane Pain, Palliative and Supportive Care Group, editor. Cochrane Database Syst Rev [Internet] 2013. Mar. 28. [cited 2019 May 1]; Available from:
http://doi.wiley.com/10.1002/14651858.CD004310.pub3.
31. Mattox TW. Cancer Cachexia: Cause, Diagnosis, and Treatment. Nutr Clin Pract 2017;Oct. 32(5):599–606.
32. Greenlee H, Balneaves LG, Carlson LE, Cohen M, Deng G, Hershman D, et al. Clinical Practice Guidelines on the Use of Integrative Therapies as Supportive Care in Patients Treated for Breast Cancer. JNCI Monogr 2014;Nov. 2014(50):346–58.
33. Cheng C-S, Chen L-Y, Ning Z-Y, Zhang C-Y, Chen H, Chen Z, et al. Acupuncture for cancer-related fatigue in lung cancer patients: a randomized, double blind, placebo-controlled pilot trial. Support Care Cancer Off J Multinatl Assoc Support Care Cancer 2017;25(12):3807–14.
34. Tao W, Luo X, Cui B, Liang D, Wang C, Duan Y, et al. Practice of traditional Chinese medicine for psycho-behavioral intervention improves quality of life in cancer patients: A systematic review and meta-analysis. Oncotarget 2015;Nov. 24. 6(37):39725–39.
35. Kim T-H, Kang JW. Acupuncture for symptoms management in Korean breast cancer survivors: a prospective pilot study. Acupunct Med J Br Med Acupunct Soc 2019;Mar. 21. 9645284198. :32610.
36. Lee Y-W, Chen T-L, Shih Y-RV, Tsai C-L, Chang C-C, Liang H-H, et al. Adjunctive traditional Chinese medicine therapy improves survival in patients with advanced breast cancer: a population-based study. Cancer 2014;May. 1. 120(9):1338–44.
38. Jeon J-H, Yoon J, Cho C-K, Jung I-C, Kim S, Lee S-H, et al. Effect of acupuncture for radioactive-iodine-induced anorexia in thyroid cancer patients: a randomized, double-blinded, sham-controlled pilot study. Integr Cancer Ther 2015;May. 14(3):221–30.
39. Grundmann O, Yoon SL, Williams JJ, Gordan L, George TJ. Augmentation of Cancer Cachexia Components With Targeted Acupuncture in Patients With Gastrointestinal Cancers: A Randomized Controlled Pilot Study. Integr Cancer Ther 2019;Dec. 181534735418823269.
40. Jeon J-H, Cho C-K, Park S-J, Kang H-J, Kim K, Jung I-C, et al. A Feasibility Study of Moxibustion for Treating Anorexia and Improving Quality of Life in Patients With Metastatic Cancer: A Randomized Sham-Controlled Trial. Integr Cancer Ther 2017;Mar. 16(1):118–25.
41. Cheon C, Yoo J-E, Yoo H-S, Cho C-K, Kang S, Kim M, et al. Efficacy and Safety of Sipjeondaebo-Tang for Anorexia in Patients with Cancer: A Pilot, Randomized, Double-Blind, Placebo-Controlled Trial. Evid Based Complement Alternat Med 2017;2017:1–9.
42. Inoue T, Takagi H, Owada Y, Watanabe Y, Yamaura T, Fukuhara M, et al. The efficacy of the Kampo medicine rikkunshito for chemotherapy-induced anorexia (RICH trial): study protocol for a randomized controlled trial. Trials 2017;Oct. 18. 18(1):485.
43. Ohnishi S, Watari H, Kanno M, Ohba Y, Takeuchi S, Miyaji T, et al. Additive effect of rikkunshito, an herbal medicine, on chemotherapy-induced nausea, vomiting, and anorexia in uterine cervical or corpus cancer patients treated with cisplatin and paclitaxel: results of a randomized phase II study (JORTC KMP-02). J Gynecol Oncol 2017;Sep. 28(5):e44.
44. Kim E, Kim H, Jang S, Kim H, Han Y, Leem J. A Cancer Patient Suffering from Chemotherapy-Induced Anorexia and Nausea Treated by Herbal Medicine (Dokhwaljihwang-tang-gamibang) Resulted in Improved Quality of Life: A Case Report. J Intern Korean Med 2018;Oct. 30. 39(5):1032–41.
46. Ahn C-B, Jang K-J, Yoon H-M, Kim C-H, Min Y-K, Song C-H, et al. A Study of the Sa-Ahm Five Element Acupuncture Theory. J Acupunct Meridian Stud 2009;Dec. 2(4):309–20.
47. Kim D-J, Park S-H, Seo J-C, Kim KS, Sohn KC, Shin IH, et al. Efficacy of saam acupuncture treatment on improvement of immune cell numbers in cancer patients: a pilot study. J Tradit Chin Med Chung Tsa Chih Ying Wen Pan 2014;Oct. 34(5):550–4.
48. Mahajan R. Real world data: Additional source for making clinical decisions. Int J Appl Basic Med Res 2015;5(2):82.