Beneficial Effects of Natural Environment Therapy for Atopic Dermatitis
Article information
Abstract
Objectives
Natural environment therapy is considered as an option for atopic dermatitis. This study was aimed to examine the beneficial effects of natural environment therapy among patients with atopic dermatitis who have moved in an atopy-free village.
Methods
The questionnaire-based survey was conducted from parents of 20 children with atopic dermatitis (12 male and 8 female, median age 8 years, ranging from 6 to 13 years). The questionnaire consisted of 10 questions for the changes of two typical symptoms of pruritus and sleeping difficulty after moving into an atopy-free village.
Results
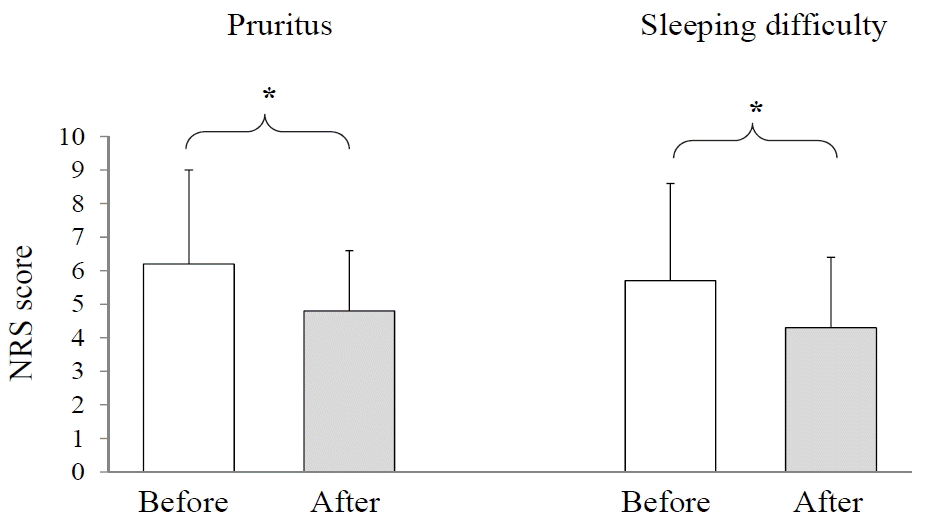
75.0% and 88.9% of patients have a family history (at least one parent and one of siblings respectively) of allergic disorders. The median staying period of patients was 27.6 months (ranging from 2 to 55 months). The score indicating pruritus was significantly improved from 6.2±2.8 into 4.8 ± 1.8 (p<0.05), and sleeping difficulty was also improved from 5.7±2.9 into 4.3±2.1 (p<0.05) too.
Conclusions
This study suggests that the natural environmental therapies could be a choice for patients with atopic dermatitis, which should be studied further for their scientific evidence and mechanisms.
Introduction
Atopic dermatitis (AD) is a chronic inflammation with the pruritic skin disorder. This disease has a high prevalence rate approximately 17.9% to 11.2% for children aged 6–14 years in Korea1), and the prevalence is continuously increasing especially in developed countries2). The etiology of AD is unclear, but the contributing factors include the genetic factor, frequent exposure to air pollutions, and westernized changes of food and housing3,4).
Atopic dermatitis seriously affects quality of life in patients as well as their families5), however no radical and effective conventional therapy exist at this moment6). The emollients, ultraviolet light exposure, and corticosteroids in severe cases are three common managements7). Therefore, adapted alternative and complementary therapies or various folk-remedies have received attention from patients with AD8). Although some herbal drugs significantly improved symptom severity of AD, still those drugs haven’t been approved as a commercial therapeutics for AD9,10).
On the other hand, it is well known that AD prevalence is higher in urban areas than rural11,12). The environmental-host interactions have influence on the risk of developing allergic diseases including AD13,14). In addition, there are reports that nature-staying atopy camp reduces the atopic symptoms and blood parameters15,16). These facts strongly propose that residence with natural environment would be an important option in management of AD.
This study was aimed to examine the beneficial effects of the natural environment therapy (termed herein for the staying in natural environments with smallest conventional therapeutics as much as possible) among patients with AD who have moved from metropolis into a designed village (called as Atopy-free village) in Geumsan of South Korea.
Participants and methods
1. Participants
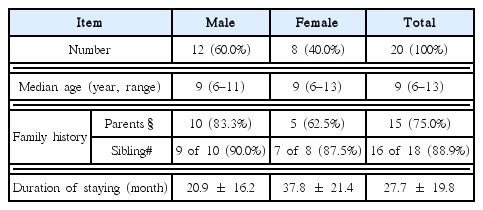
All kindergarten and elementary schoolchildren who have been staying in Atopy-free village, and previously diagnosed with AD and had the duration of staying > one month at Atopy-free village participated in this study. Total 20 participants consisting of 12 male (median age 9 year, ranging from 6~11 years) and 8 female (median age 9 year, ranging from 6~13 years) were enrolled.
2. Methods
A questionnaire was given to the mothers of 20 children participants between July to August 2016. The questionnaire consisted of 10 questions for the demographic information, family history, and duration of staying at Atopy-free village as well as changes of two main AD symptoms, pruritus and sleeping difficulty before the movement and during the recent month respectively. For quantitative assessment of pruritus and sleeping difficulty, a numerical self-rating scale (NRS) was used; mothers scored each item from 1 to 10 (1 indicating no symptom versus 10 indicating endurable status). All participants’ parents fully comprehended the purpose of the study, and provided written informed consent.
3. Atopy-free village
The Atopy-free village was founded at by Geumsan city, a Korean local government in 2010. This village is located at Sanggok-ri Gunbook-myun Geumsa-gun South Korea, surrounded by multiple mountains including Seadae Mountain with 978m height. The concept of Atopy-free village is composed of chemical-low environments including mud-timber buildings with environment-friendly materials and organic food in housing as well as in school life. The kindergarten and elementary schoolchildren were educated with mainly outdoor activities relatively other than schools in city.
4. Statistical analysis
The data were analyzed whether pruritus and sleeping difficulty were improved after moving into Atopy-free village comparing to before. The paired t-test was conducted for statistical analysis using PASW Statistics 17 program, and if p value was less than 0.05, it was considered as statistically significant.
Results
1. Characteristics of participants
The average duration of staying at Atopy-free village was 27.7±19.8 months (male 20.9±16.2 and female 37.8±21.4 months). Seventy-five percent of children had at least one parent with allergic disorders including AD, allergic rhinitis or asthma respectively. In case they had brother or sister, 88.9% of children had a sibling with one of above allergic disorders (Table 1).
2. Improvement of pruritus and sleeping difficulty
The NRS average score for pruritus was 6.2±2.8 (male 7.2±2.4 and female 4.6±2.8), whereas the pruritus severity was significantly improved into 4.8±1.8 (p<0.05, male 4.9±2.1 and female 4.5±1.4) respectively. The sleeping difficulty was also improved significantly from 5.7±2.9 (male 7.1±2.3 and female 3.8±1.8) to 4.3±2.2 (p<0.05, male 4.6± 2.4 and female 3.5±2.5) respectively (Fig. 1).
Discussion
AD has profound impacts on patient and family quality of life, however no satisfactory preventive or treatment strategy exist. The exploration of linkage between modern urban life and various diseases resulted in attending natural environments as one of therpeutics17). Several Korean studies presented that forest activity-based atopy camps reduced AD symptoms and induced immunomodulation18,19). One survey study in Korea reported that many parents having children suffering from AD expected the beneficial effects of natural environment therapy, and 55.2 % of parents had considered moving to a rural area20).
In order to get a clue for the clinical benefit of staying in natural environments without any conventional therapeutics as much as possible (termed as natural environment therapy in this study), the present study examined the improvement of AD symptoms. The average residence period at Atopy-free village was 27.7±19.8 months (ranging from 2 to 55 months, Table 1). The pruritus is a typical symptom of AD, and it usually becomes worse at night, resulting in sleep disturbance21). The changes of pruritus and sleeping status therefore could be proper parameters for clinical assessment in AD patients. In the present study, parents reported that both pruritus and sleeping difficulty significantly reduced after movement into Atopy-free village (Fig. 1). AD generally occurs in children, which impairs the emotional and intellectual development, and then be very stressful to their parents22). Accordingly, the assessment by parents in current study would be convincing. In fact, half of AD is known to disappear in cases of early onset and mild severity, however likely present study cases, family history of atopy and severe AD are associated with poor prognosis23). The patients in our study showed high rate of family history for allergic disorders; 75% of parents as well as 88.9% of sibling (Table 1).
The Atopy-free village was founded by Geumsan city, a Korean local government. The basic strategy is living within natural environments at a countryside surrounded by mountains, and mud-timber buildings and organic food in housing and school life. The relationship between diets with incidence of AD is well known, and then organic food is believed to reduce the risk of AD24,25). All patients in present study had moved into the village from metropolises for their severe AD. They were not satisfied with conventional therapies, and quitted them. The uncontrolled AD impairs the life’s quality of patients as well as their families26,27), which they would sometimes prefer to move to a countryside.
Above findings proposes the potential of environmental therapy for children with AD. The forest environment-based programs showed partially positive effectiveness in the management in alcoholics and elderly hypertensive patients28,29). Our previous studies also observed the reduction of pruritus in children with AD during short-term environment-based atopy camp and long-term residence in Atopy-free village15,30). For assessment of the changes of AD symptoms, above studies adapted the objective tools and parameters such as score of SCORAD and EASI or serum IgE concentration respectively. In present study, the parents assessed the changes of two typical AD symptoms, pruritus and sleeping difficulty. This would be a week point of our study along with the relatively small number of participants. The parents’ assessment after observation for long term however could present the alternative evaluation of AD symptoms comparing to an examination at certain time point, because AD-related symptoms and biomarkers would be changeable depending on the time points.
Taken together, this study provides important clinical information for development of the natural environment therapy for parents of AD. There are, however, still large tasks to be done, including multiple scientific studies to extend evidence for clinical effect as well as its mechanisms.
Acknowledgments
This research was supported by the “Leaders Industry-university Cooperation” Project, supported by the Ministry of Education, Science & Technology (2016B-0016-010107), and the grant of the Traditional Korean Medicine R & D Project, Ministry of Health & Welfare, Republic of Korea (HI16C-0112-000015).
Notes
Conflicts of interest
The authors declare that there is no conflict of interests.