References
1. Choie MH, Jeong IS, Jeong TY, Kim KS, Yoo HS. Effects of reflexology foot massage to improve quality of life and reduce anxiety for hospitalized cancer patients undergoing chemotherapy or radioisotope therapy. J Korean Trad Oncol 2009;14:61–74.
2. Windebank AJ, Grisold W. Chemotherapy -induced neuropathy. J Peripher Nerv Syst 2008;13(1):27–46.
3. Wolf S, Barton D, Kottschade L, Grothey A, Loprinzi C. Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. European journal of cancer 2008;44(11):1507–15.
4. Housheer FH, Schilsky RL, Bain S, Berghorn EJ, Lieberman F. Diagnosis, management, and evaluation of chemotherapy-induced peripheral neuropathy. Emin Oncol 2006;33:15–49.
5. Ajani JA, Welch SR, Raber MN, Fields WS, Krakoff IH. Comprehensive criteria for assessing therapy-induced toxicity. Cancer Invest 1990;8:147–59.
6. Miller AB, Hoogstraten B, Staquet M, Winkler A. Reporting results of cancer treatment. Cancer 1981;47:207–14.
7. Oken MM, Creech RH, Tormey DC, Horton J, Davis TE, McFadden ET, et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol 1982;5:649–55.
8. Cavaletti G, Jann S, Pace A, Plasmati R, Siciliano G, Briani C, et al. Multi-center assessment of he Total Neuropathy Score for chemotherapy-induced peripheral eurotoxicity. J Peripher Nerv Syst 2006;11:135–41.
9. Kwak MK, Kim EJ, Lee ER, Kwon IG, Hwang MS. Characteristics and Quality of Life in Patients with Chemotherapy-Induced Peripheral Neuropathy. Journal of Korean oncology nursing 2010;10(2):231–9.
10. Berger T, Malayeri R, Doppelbauer A, Krajnik G, Huber H, Auff E, et al. Neurological monitoring of neurotoxicity induced by paclitaxel/cisplatin chemotherapy. Eur J Cancer 1997;33(9):1393–9.
11. Forsyth PA, Balmaceda C, Peterson K, Seidman AD, Brasher P, DeAngelis LM, et al. Prospective study of paclitaxel-induced peripheral neuropathy with quantitative sensory testing. J Neurooncol 1997;35(1):47–53.
12. Du Bois A, Schlaich M, Luck HJ, Mollenkopf A, Wechsel U, Rauchholz M, et al. Evaluation of neurotoxicity induced by paclitaxel second -line chemotherapy. Support Care Cancer 1999;7(5):354–61.
13. Kim JH, Choi KS, Kim TW, Hong YS. Quality of life in colorectal cancer patients with chemotherapy-induced peripheral neuropathy. Asian Oncology Nursing 2011;11(3):254–62.
14. Kim HY, Kang JH, Youn HJ, So HS, Song CE, Chae SY, et al. Reliability and validty of the Korean version of the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire to assess chemotherapy -induced peripheral neuropathy. J Korean Acad Nurs 2014;44(6):735–42.
15. Tofthagen CS, McMillan SC, Kip KE. Development and psychometric evaluation of the chemotherapy-induced peripheral neuropathy assessment tool. Cancer Nurs 2011;34(4):E10–20.
16. Usha KM, Jayantee K, Pradeep PN. Diagnostic approach to peripheral neuropathy. Ann Indian Acad Neurol 2008;11(2):89–97.
18. Seyfried Thomas N, Flores Roberto E, Poff Angela M, D’Agostino Dominic P. Cancer as a Metabolic Disease. Carcinogenesis 2014;35(3):515–27.
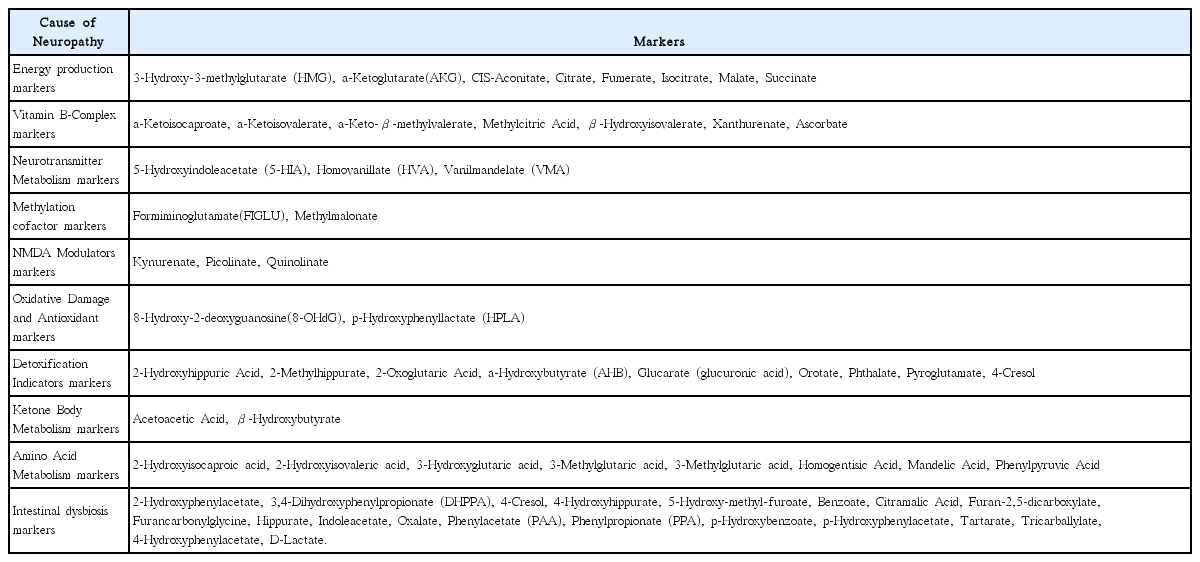
19. Lord RS, Bralley JA. Laboratory evaluations for integrative and functional medicine Duluth. GA: Metametrix Institute; 2008.
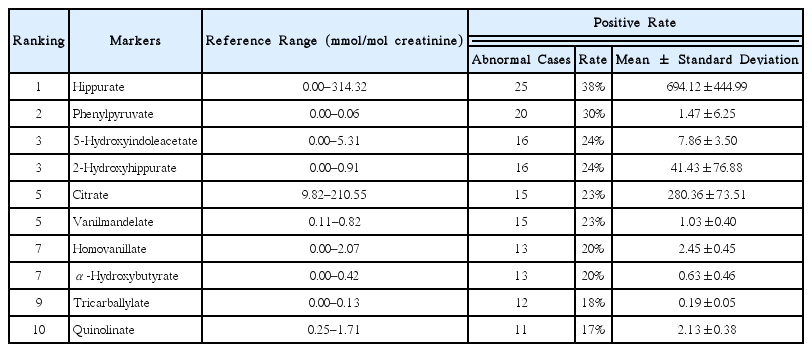
20. Orgacka h, Zbytniewski Z. Excretion of vanillic acid and homovanillic acid and tissue distribution of catecholamines and their metabolites in mice with various levels of pigmentation. Endocrinol Pol 1991;42(3):471–9.