Introduction
Immunity is an important and complex component of the defense mechanism of the body that comprises innate and adaptive immunity1). Immune function refers to the improvement of biomarkers related to the enhancement of immune function and alleviation of irritable immune response. Immunity-enhancing functionality refers to a function that enhances the ability to protect the body by regulating immunity. The function of alleviating an overactive immune response refers to a function for suppressing an undesirably increased immune response, such as a reaction to an allergic reaction autoantigen or a modified autoantigen caused by a reaction to an external substance2). Traditional oriental medicines have been widely used in alleviating immune-related diseases in the Asian population for a long time. There has been significant progress in research focusing on the enhancement of immunity using natural materials over the recent years. Ginseng and red ginseng have been used extensively for immune enhancements in Korea3,4). Chemotherapeutics inhibit the growth of cancer cells, but they can damage normal cells or cause side effects, such as immunosuppression5). Moreover, the increase in particulate dust matter and other forms of environmental pollution results in specific diseases. Therefore, the regulation of immune response plays an important role in the prevention and treatment of diseases. Macrophages are multifunctional cells that are crucial in innate and adaptive immunity6). During pathogen invasion, the adaptive immune system cooperates with innate immunity to eliminate pathogenic microorganisms7).
Lipopolysaccharide (LPS) activates macrophages. LPS binds to toll-like receptor 4 (TLR4) that primarily directs the immune response by activating the macrophages and secreting cytokines8). Activated macrophages secrete a variety of cytokines and chemokines, such as interferon-γ, interleukin (IL)-1β, IL-6, IL-12, tumor necrosis factor (TNF)-α, nitric oxide (NO), and prostaglandin E29). Thus, it is imperative to understand macrophage-mediated immunity.
Pueraria lobata belongs to the Leguminosae family. The roots and flowers of P. lobata are widely used in traditional medicine10,11) primarily for cardiovascular diseases, hangover, inappetence, and vomiting11,12). Previous studies have shown that the active compounds of P. lobata extract comprise isoflavones, including kakkalide, kakkalidone, puerarin, irisolidone, 6″-O-xylosyl-glycitin, tectoridin, 6″-O-xylosyl-tectoridin, and genistin13,14,15).
Most studies on the extract of P. lobata have focused on its effects as an antioxidant, anti-inflammatory agent, and hangover remedy 10,11,16). However, the immune activity of the extract remains to be studied. Therefore, we wanted to understand the immune effects of the extract of P. lobata flowers. In this study, we report the immuopotentiating effects of the P. lobata flower extract (PLFE) on RAW264.7 cells based on the production of NO and cytokines and NF-κB/MAPK signaling.
Materials and Methods
1. Materials
Lipopolysaccharide (LPS), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma Chemical Co. (St. Louis, MO, USA). P. lobata flowers were obtained from Hanbang Herbal Drug Co. (Jecheon, Korea). Bradford reagent was procured from BioRad (Hercules, CA, USA). Antibodies against iNOS and COX-2 were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA); antibodies against IκBα, p-IκBα, p-p38, p-JNK, and p-ERK were obtained from Cell Signaling Technology (Beverly, MA, USA). All other reagents were of the highest grade commercially available.
2. Preparation of PLFE
PLFE was prepared as described. Briefly, dried P. lobata flowers (100 g) were extracted with distilled water. The water extract was obtained by refluxing the sample for 90 min at room temperature followed by suction filtering through filter paper (Whatman No. 41). The filtered extracts were evaporated under reduced pressure using a rotary vacuum evaporator (EYELA, Tokyo, Japan) at 50°C. After evaporation, the water and ethanol extracts were lyophilized using a freeze dryer (Samwon Industry, Seoul, Korea).
3. Cell culture
Murine macrophage RAW264.7 cells were purchased from the Korean Cell Line Bank (Seoul, Korea) and maintained in high glucose Dulbecco’s modified Eagle’s medium supplemented with 10% heat-inactivated fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C in a humidified incubator (5% CO2 and 95% air).
4. Cell viability
The cytotoxic effects of the extracts on RAW264.7 cells were evaluated using the MTT assay. Briefly, RAW264.7 cells were seeded in 96-well plates at a density of 2×104 cells/well. Cells were treated with 0, 25, 50, or 100 μg/mL of PLFE or LPS (100 ng/mL) for 24 h. The cells were then incubated with MTT solution for 2 h. Subsequently, the supernatant was removed and resulting formazan crystals were dissolved in dimethyl sulfoxide before absorbance at 540 nm was measured using a microplate reader (SpectraMax M2/M2e, Molecular Devices, Sunnyvale, CA, USA).
5. Measurement of NO generation
NO levels in the cell culture medium were analyzed by determining the accumulation of nitrite in the cell culture medium using the Griess reagent and sodium nitrite as the standard. RAW264.7 cells were treated with PLFE for 18 h before the culture medium (100 μL) was transferred into each well of a 96-well plate. An equal volume (100 μL) of Griess reagent was added to each well and incubated for 10 min at room temperature. Finally, the absorbance at 550 nm was measured using a microplate reader (SpectraMax M2/M2e, Molecular Devices, Sunnyvale, CA, USA).
6. Inflammatory cytokine production
RAW264.7 cells (1×105 cells/well) were seeded into 24-well plates and incubated overnight for cell adherence. After treatment with PLFE or LPS (positive control) for 18 h, TNF-α and IL-6 levels in the cell supernatants was assayed using enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions.
7. Western blotting
Treated cells were harvested, lysed, and subjected to protein quantification using the Bradford assay. Samples containing 15–20 μg of proteins were subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (BioRad Laboratories, Hercules, CA, USA) and separated proteins were transferred to polyvinylidene fluoride membranes (GE Healthcare, Buckinghamshire, UK). After blocking with 5% skim milk in Tris-buffered saline supplemented with Tween-20 for 1 h at room temperature, the membranes were incubated overnight with specific primary antibodies at 4°C. After washing thrice with the Tris-buffered saline with Tween-20, the membranes were incubated with species-specific horseradish peroxidase-conjugated secondary antibodies for 1 h at room temperature. Protein bands were developed using enhanced chemiluminescence reagents and the immunoblots were visualized using the Davinch-Chemi™ imaging system (Young Wha Scientific Co. Ltd, Seoul, Korea). Densitometry for the target protein levels were normalized to β-actin.
8. Statistical analysis
The results are expressed as the mean ± standard deviation (S.D.) values. Statistical analysis was performed by analysis of variance (ANOVA), followed by Dunnett’s test to determine the significance of differences using GraphPad Prism 5.0, p < 0.05 was considered statistically significant.
Results
1. Effect of PLFE on cell viability and NO production in RAW264.7 cells
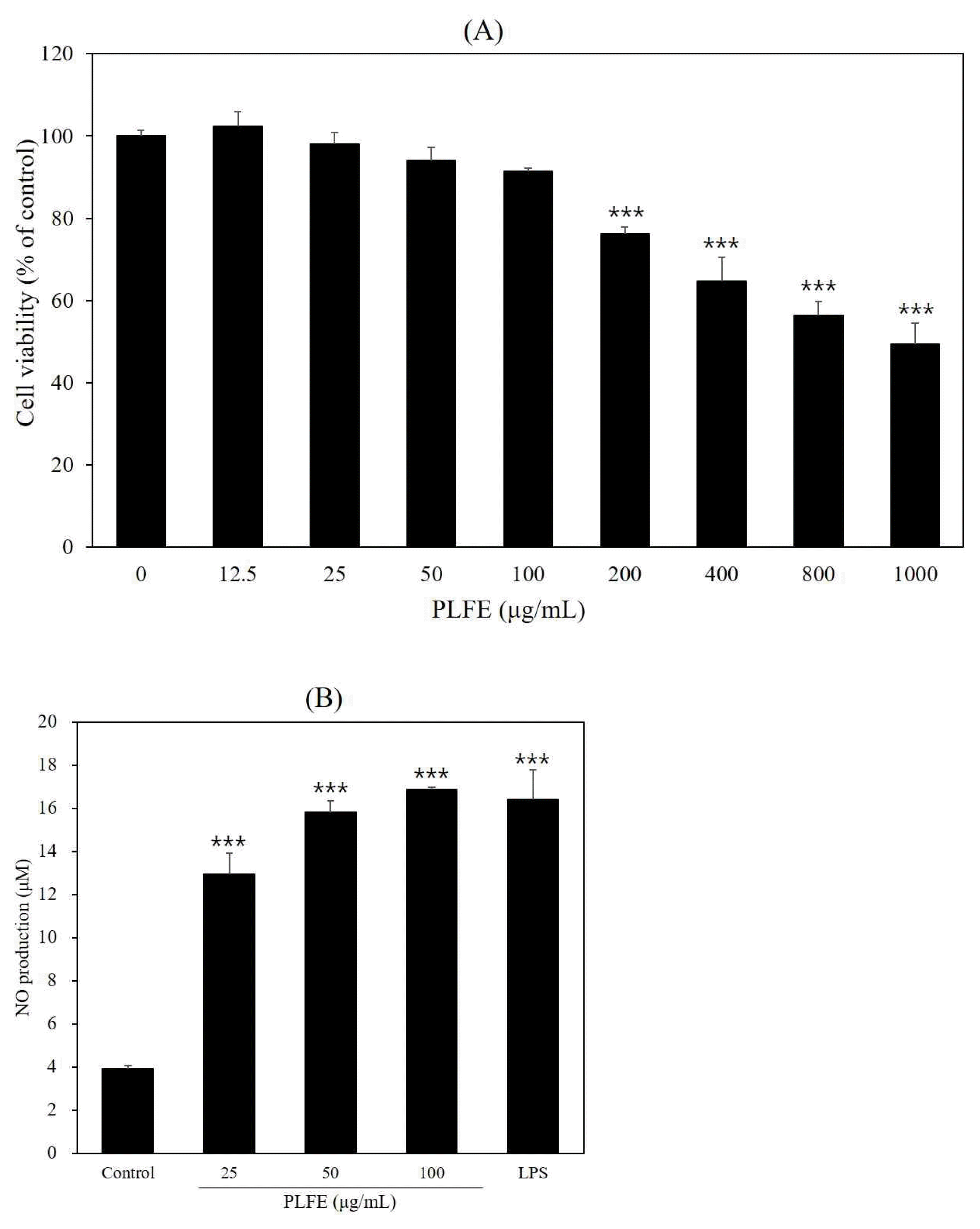
To determine the effect of PLFE on the cell viability, cells were treated with various concentration of PLFE (0–1,000 μg/mL). PLFE showed over 90% of cell viability below 100 μg/mL concentration and didn’t showed toxicity (Fig. 1A). Therefore, we performed the following experiments with the experimental concentration of 100 μg/mL as the highest concentration. To assess the enhanced-immune effect of PLFE, we examined whether PLFE affected the NO production in RAW264.7 cells. As shown Fig. 1B, PLFE increased the NO production in dose-dependent manner. Compare with the control group, NO production was markedly increased (p<0.001) the treatment of PLFE as much as LPS group. Thus, only PLFW was subjected to further analysis to explore its immuno-stimulating effect and mechanisms.
2. Effect of PLFE on the expressions of TNF-α and IL-6
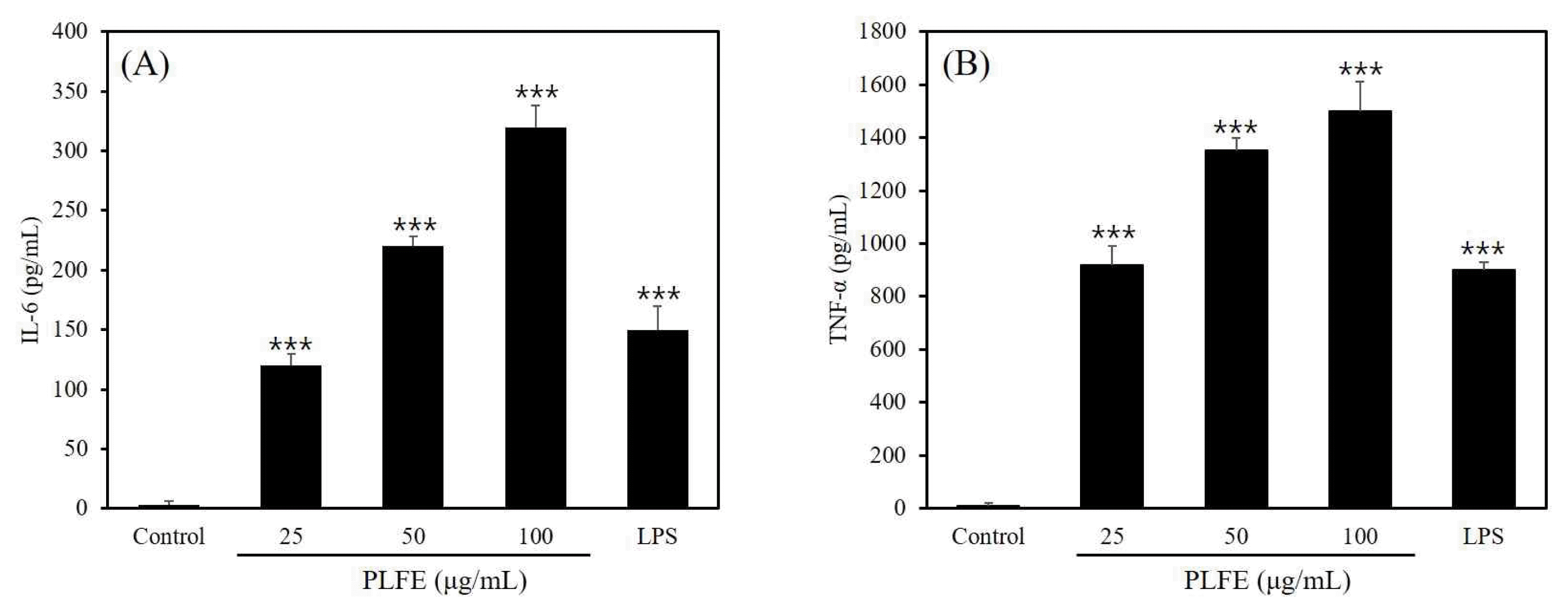
Cytokines are important mediators involved in modulating immune response and inflammatory reactions, especially during infection and trauma 17). The cytokines, including TNF-α and IL-6, are potent immunomodulators in activated macrophages. Thus, we determined if PLFE affected the expression of cytokines in RAW264.7 cells. As shown in Fig. 2, compared with the control group, IL-6 and TNF-α levels increased in the PLFE-treated group (p<0.001). The amount of cytokines secreted from PLFE-treated cells was higher than that from LPS-treated cells (positive control). These results suggest that PLFE promotes immunostimulation in RAW264.7 cells.
3. Effect of PLFE on iNOS and COX-2 expression
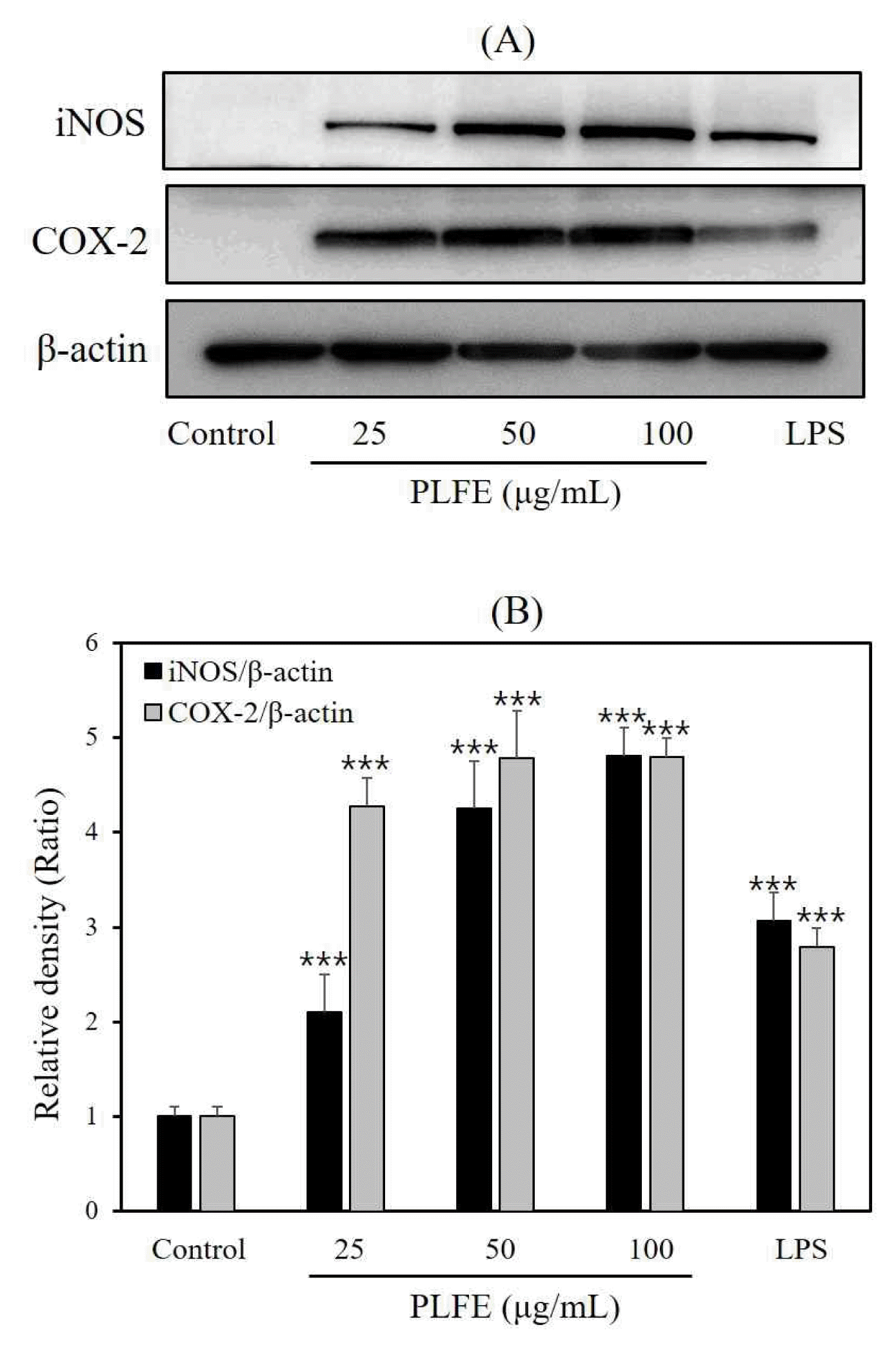
To further demonstrate the immune stimulatory effect of PLFE, we determined the protein levels of iNOS and COX-2 using western blotting. As shown in Fig. 3, the protein levels of iNOS and COX-2 increased in PLFE-treated cells in a dose-dependent manner. Cells treated with various concentrations of PLFE (25, 50, and 100 μg/mL) for 18 h significantly increased iNOS and COX-2 protein levels compared to those in the control. We used LPS as a positive control to show the strong expression of iNOS and COX-2 compared to the control sample. In addition, PLFE induced the expression of iNOS and COX-2 stronger than that observed in LPS-treated cells. Thus, PLFE may upregulate pro-inflammatory mediators, such as iNOS and COX-2.
4. Effect of PLFE on IκBα degradation
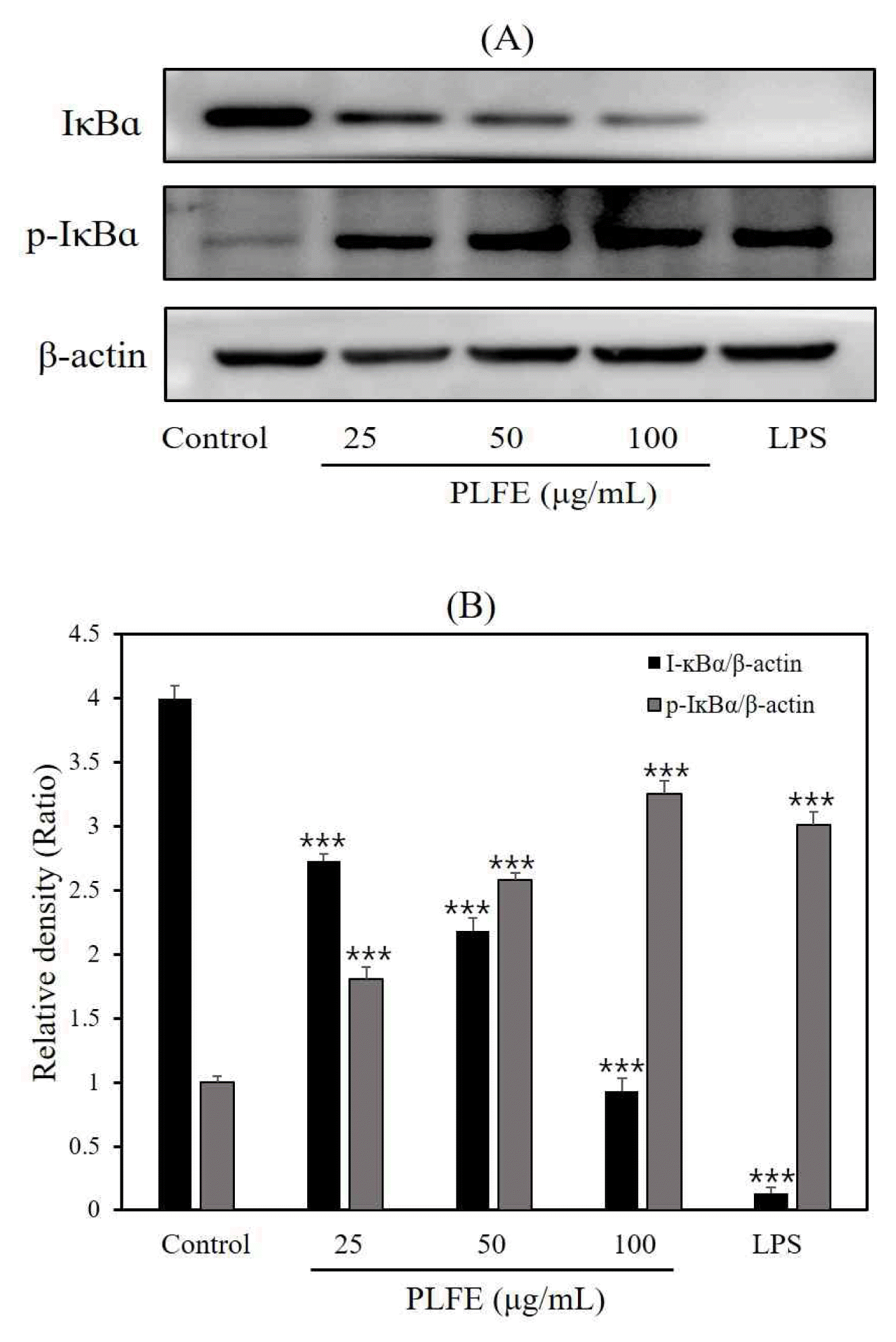
IκBα degradation is crucial in the activation of the transcription factor NF-κB. Thus, we investigated the effect of PLFE on IκBα degradation: IκBα protein levels decreased in PLFE or LPS-treated cells (Fig. 4A, 4B). In addition, PLFE increased the phosphorylation of IκBα. IκBα constitutes a family of cellular proteins that inhibit NF-κB activity by masking the nuclear localization signal of NF-κB proteins and sequestering their inactive forms in the cytoplasm18). External stimuli that activate macrophages enhance IκBα phosphorylation, leading to its degradation and simultaneous activation and nuclear translocation of NF-κB to bind to its cognate DNA-binding site in the regulatory region of multiple genes19). Therefore, our results suggested that PLFE activates NF-κB, thereby enabling the target gene transcription in macrophages.
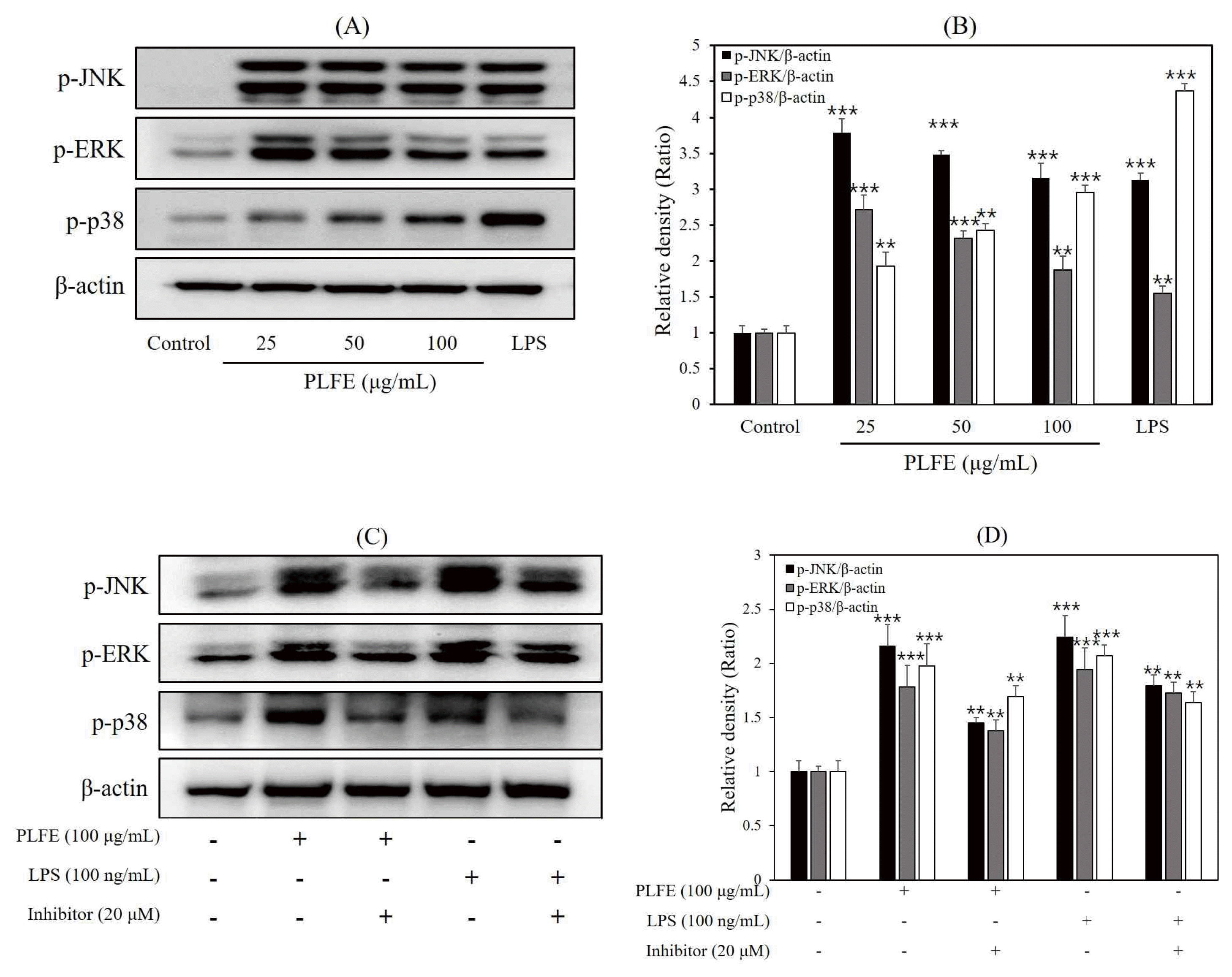
5. Effect of PLFE on MAPK signaling
To investigate whether PLFE affects immunity via MAPK signaling, we evaluated the protein levels of the MAPK family of proteins, including p38, JNK, and ERK1/2 using western blot analysis. As shown in Fig 5A, 5B, 30 min of PLFE and LPS increased the phosphorylation of MAPK isoforms, p38, JNK, and ERK1/2. In addition, MAPK expression was inhibited in cells treated with PLFE and MAPK-specific inhibitor (Fig. 5C, 5D). Taken together, PLFE stimulated MAPK signaling, thereby upregulating iNOS and COX-2 by activating NF-κB.
Discussion
Macrophages comprise an integral component of the first line of defense in protecting the host from infection20) by eliciting immune responses 21). Macrophages are one of the important innate immune cells that make up our body, and are distributed in various forms in all tissues, and under normal conditions, they play a role in protecting the body through the predation of invading foreign pathogens and toxic substances.
Since abnormalities in the function of macrophages can cause various diseases, research on macrophages is being actively conducted. M1 macrophages are a type of macrophage with typical functions, and M2 macrophages are alternative activated macrophages with a wide range of physiological pathological effects. M2 macrophages have different functions than M1 macrophages, are important for cell homeostasis and inflammatory reactions, and have specific effects on tissue repair or metabolic processes. Research on M2 macrophages has been actively conducted in recent years, and various subtypes have been discovered and reported. It is polarized by specific substances or various kinds of reactions and is subdivided into new types of M2 macrophages such as M2a, M2b, M2c, and M2d22). In addition, it has been reported that various types of macrophages have an important influence on not only immune function but also metabolic regulation23). Contrary to the common knowledge that immune cells will suppress cancer, studies have also been reported on macrophages that promote tumor development, such as Tumor-associated macrophage (TAM)24). NO is a signaling molecule that is secreted by macrophages, neurons, and endothelial cells and function in protecting or being toxic to cells under specific conditions25,26). This study showed that PLFE increased the production of NO by upregulating iNOS in macrophages. We concluded that PLFE regulated the synthesis of pro-inflammatory cytokines in RAW264.7 cells.
Cytokines are secretory proteins from immune and interstitial cells involved in intercellular communication and exchange of biomolecules. Cytokine function is classified based on their pro-inflammatory or anti-inflammatory effects and associated signal transduction is involved in the regulation of immune response27,28). IL-10 is an anti-inflammatory cytokine, while IL-6 and TNF-α are pro-inflammatory cytokines that comprise a key area of research in immunology 29).
IL-6 is a soluble protein with pleiotropic effects on inflammation, immune response, and hematopoiesis30) and is secreted from numerous cells, including T cells, B cells, mononuclear phagocytes, fibroblasts, hepatocytes, endothelial cells, keratinocytes, and bone marrow cells31). IL-6 stimulates T cell proliferation and, together with IL-4, participates in the generation of the Th2 immune response32,33). IL-6 also participates in triggering humoral immunity by stimulating B cell differentiation and antibody secretion34).
TNF-α is a central regulator of inflammation produced by neutrophils, lymphocytes, and natural killer, endothelial, and mast cells35). The soluble active form of TNF-α is cleaved from its membrane-bound form using the TNF-α converting enzyme36). TNF-α and TNF-β induce antitumor immunity via direct cytotoxic effects on cancerous cells37). In this study, we investigated the immune-enhancing activity of PLFE based on cytokine production in murine macrophage cells. We have demonstrated that PLFE enhanced the synthesis of IL-6 and TNF-α in a dose-dependent manner. PLFE may exert its immunostimulatory effect by increasing the secretion of NO and cytokines. MAPK (p38, JNK, and ERK) signaling is upstream of IL-1β, IL-6, and TNF-α synthesis38,39). PLFE-treated RAW264.7 cells showed an upregulation of MAPK pathway components. In order to confirm the immunostimulatory effect, IL-6 and TNF-a were measured, and IL-10, a deduction inhibitory cytokine, was not measured, but it will have to be confirmed in further experiments.
NF-κB is a transcription factor for immune response-related gene expression and plays an important role in the immune function of monocytes and macrophages40). IκBα inhibits NF-κB by masking its nuclear localization signal and retaining an inactive form of NF-κB in the cytoplasm18). This study showed that PLFE decreased IκBα and increased the phosphorylation of IκBα. Therefore, PLFE activated NF-κB by degrading IκBα. In order to find out this mechanism more clearly, the expression of NF-kB and p-NF-kB should be confirmed.
According to the health functional food functional evaluation guide proposed by the Ministry of Food and Drug Safety, the function related to the improvement of immune function is evaluated in terms of two aspects: enhancing immune function and helping to alleviate hypersensitivity immune response2). This study was designed to improve immune function, and analysis of the components of PLFE and animal experiments should be conducted in the future.
Conclusions
We analyzed the effects of PLFE on immune regulation in RAW264.7 cells. PLFE enhanced the immune activity of the murine macrophage RAW264.7 cells by activating NF-κB/MAPK signaling, thereby increasing the secretion of NO and cytokines (IL-6 and TNF-α). In summary, we have identified the main components involved in the immunostimulatory and immune enhancement effects of PLFE using an in vivo model.