Introduction
The rate of liver cancer in Korea is estimated at 30.5 per 100,000 men and 7.6 per 100,000 women, and the third annual report on the Korea Central Cancer Registration Project showed that it was the seventh for men and the seventh for women, which was higher than for other OECD countries1),2).
The mistletoe is a plant parasitic on shrubs that has long been used as a medicinal plant, and attempts to use it in malignant tumors have recently been made using the extract3).
Mistleto extracts are known to cause changes in the human immune system, and because of these immune-control effects, malignant tumors such as malignant lymphoma, multiple myeloma, leukemia are treated, malignant tumors are intended to prevent recurrence after primary treatment, or pre-cervative tumors such as chronic hepatitis B, and primary bone treatment of patients4).
The anti-cancer effects on Be Venom were actively implemented both at home and abroad, and the effects of cancer were reported due to the ingredients of Bong-dock, immunotherapy, pain, and so on. Be Venom is peptide components (melithin, adolapin, MCD sensitive, etc.), enzymes (PLA2, Hyaluronidase, Glucosidase, etc.), amines (histamin, epinephrine, etc.), non-ptitious components Beef poison is 1.3 percent, ph 5.2 percent bitter, weak-tasting, and 75 percent protein5).
In this study, we report that mistletoe and Be Venom were carefully studied to induce apoptosis of Hep G2, the liver cancer cell owner.
Material and Methods
1. Preparations of bee venom
1g of the poison Apis Melipera ligustica (West honey bee, Sigma, USA) was dissolved in the poison distilled water.
The mistletoe was purchased from Viscum album colorectum agglutin, VCA (Related per Cheongun, South Gyeongsang and South Korea) and used as the extraction method below.
2. Method
1) Cellular stock and cell culture
The hepatoma cell line and the Hep G2 cell used in the experiment were purchased from the American Type Culture Collection (Rockville, MD). Cells were incubated using Dulbecco’s modified Eagle’s Medium (DMEM, Invitrogen, USA) containing 10% Fetal Bovine Serum (FBS; Invitrogen, USA) at 37°C and 5% CO2 incubator. To prevent contamination, 100ut/ml phenicillin, 100ug/ml streptomin (Gibco/BRL, USA) was added as an antibiotic. Trypsin-EDTA (Gibco/BRL, USA) was treated and stratified. The badge was exchanged every two or three days.
2) Cellular activity measurement (MTT allay)
MTT ((4,5-dimethylthiazol-2-yl)-2,5-diphenyltrazolium bromide, Sigma) alay was conducted using Sladowski’s method. MTT assay is a test method that utilizes the ability of cells to reduce MTT titrazol, a yellow soluble substrate, to a green-colored MTT FORMazan by dehydrogen enzyme. The absorption of MTT formazan is greatest at a wavelength of 580nm, where the absorbance measured is reflected in the oxidative power of a cell that is alive and has a strong metabolism6).
MTT assembly was first incubated for 24 hours after 16 hours of Hep G2 cells (5x103 cells/well) in a 12 cell plate. And 3-(4-Dimethylthiazol-2-yl)-2,5-diphenyltetra-zolium bromide (MTT). Sigma) 2 mg/ml was added, incubated in the incubator for 4 hours, then dissolved with dimethyllsulfoxide (DMSO; Sigma) and measured absorbency with a microplate reader (Molecular devices, USA) at a wavelength of 580nm to calculate cell survival.
Cell viability (%) is defined as: If the value of the normal range is controlled and the O.D. value is defined as 100 percent survival of the cell, and the value of the O.D. measured by the rest of the group is calculated as follows.
3) Medication of inhibitors
PH3K inhibitor (LY2942), p38 MAPK inhibitor (SB203580), MAPK inhibitor (PD98059), and JNK inhibitor (SP600125) (Calbiochem, USA) treated the extraction and sterilization rates before the end of 30 minutes and then treated them with the MTT method.
4) Western blot analysis method
All cell solvents were made by boiling cells within a sample buffer (66.5 mmol/1 Tris-HCl, pH 6.8, 2% SDS, 20% glycerol, 10% 2-mercaptoethanol). Protein doses were used in the bicinchonic acid (BCA, Pierce) method. 50 ug of the quantified protein sample was separated by 4–12% sodium diecylsulfate-polyacrylamide gradient gel (Invitrogen) and transferred to the nitrocellulose paper (Amersham). It was confirmed that the protein was completely transferred by dyeing the membrane of protein with Ponceau-S. After washing it with a Tris-buffered saline (TBS-T) containing 0.1% Tween 20, the protein was blocked with 5% de-fat milk solution for more than 30 minutes. p38 (p-p38), p38, MAPK (p-MAPK), MAPK, ERM (p-EMM), moesin, JNK (p-JNK), JNK antibodies, PARP-1, Bcl-x, and activated (Cell signing) are in the USAch After the second antibody reaction, the curtain was washed and the protein that they wanted was visualized through the advanced chemical protection system (ECL, Pierce). Visualization and quantitative analysis of proteins was done using image equipment (LAS-3000, Fujitsu
5) DNA fragmentation
10 ug/ml of BV and 50 ug/ml of VCA beef were processed in Hep G2 cells (5x105 cells), then cells were recovered for 16 hours and 24 hours respectively, centrifugation was performed for 5 minutes at 800 rpm, 10 mM Tris (pH7.4), 10 m EDTA (pH 8.0), and the floating solution was filled with a buffer of 10 mM (X00-1). Add 20 mg/l of RNase A and 20 mg/l of Proteinase K to react for another hour at 37 °C and centrifugation to obtain genomic DNA. Add TE buffer (10mM Tris (pH7.4) and 10mM EDTA (pH 8.0) to dissolve DNA and electro-activate the 1% agarose gel to check the band.
6) FACS Analysis
After processing 10 ug/ml of BV and 50 ug/ml of VCA milk fat in the Hep G2 cell (2x106 cells), recover the cells each 24 hours, wash them with cold PBS, and secure them at room temperature for 30 minutes. Then, add 0.05 mg/ml of RNase A to remove RNA and react for one hour at 37°C. Then, add 50g/ml of Propium iodide (PI) to the specimen and react in the dark for 30 minutes. DNA analysis used ELITE ESP flow rateometer (Bekman-Coulter Inc., USA).
7) Data analysis and statistical processing
All measurements were expressed as the mean value standard deviation (mean ± S.D.) and statistical analysis among each group of experiments performed one-way ANOVA of the SPSS program for windows, and post-verification was verified by Tukey test. The statistical significance of the whole experiment was recognized as significant if the p value was less than 0.05.
Result
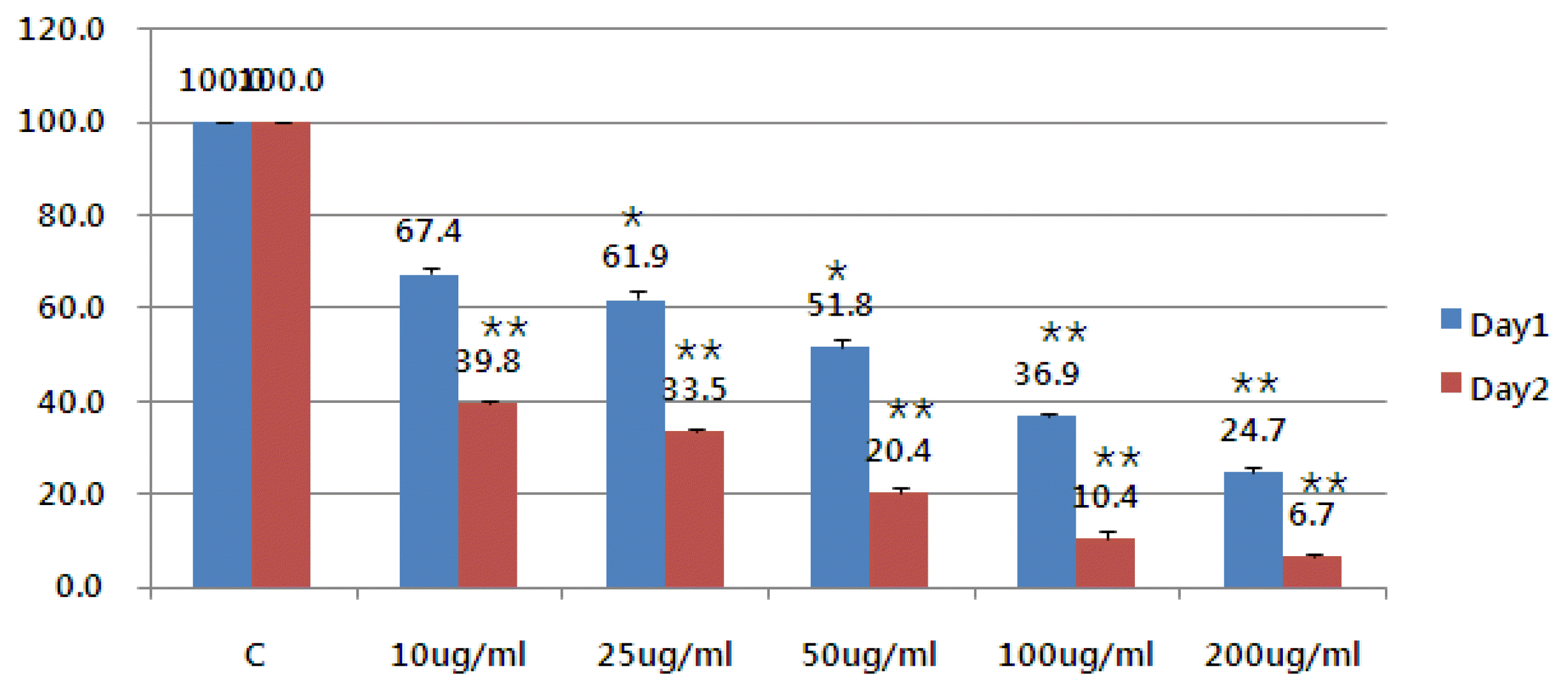
1. Effect of Mistleto (VCA) on the survival rate of liver cancer cells
The cell survival rate was observed 24 hours and 48 hours after mistletoe was treated by concentration to investigate the effect of mistletoe on the survival rate of liver cancer cells. Concentration-dependent degradation from 10 ug/ml to 200 ug/ml resulted in increased apoptosis after 48 hours (Figure 1).
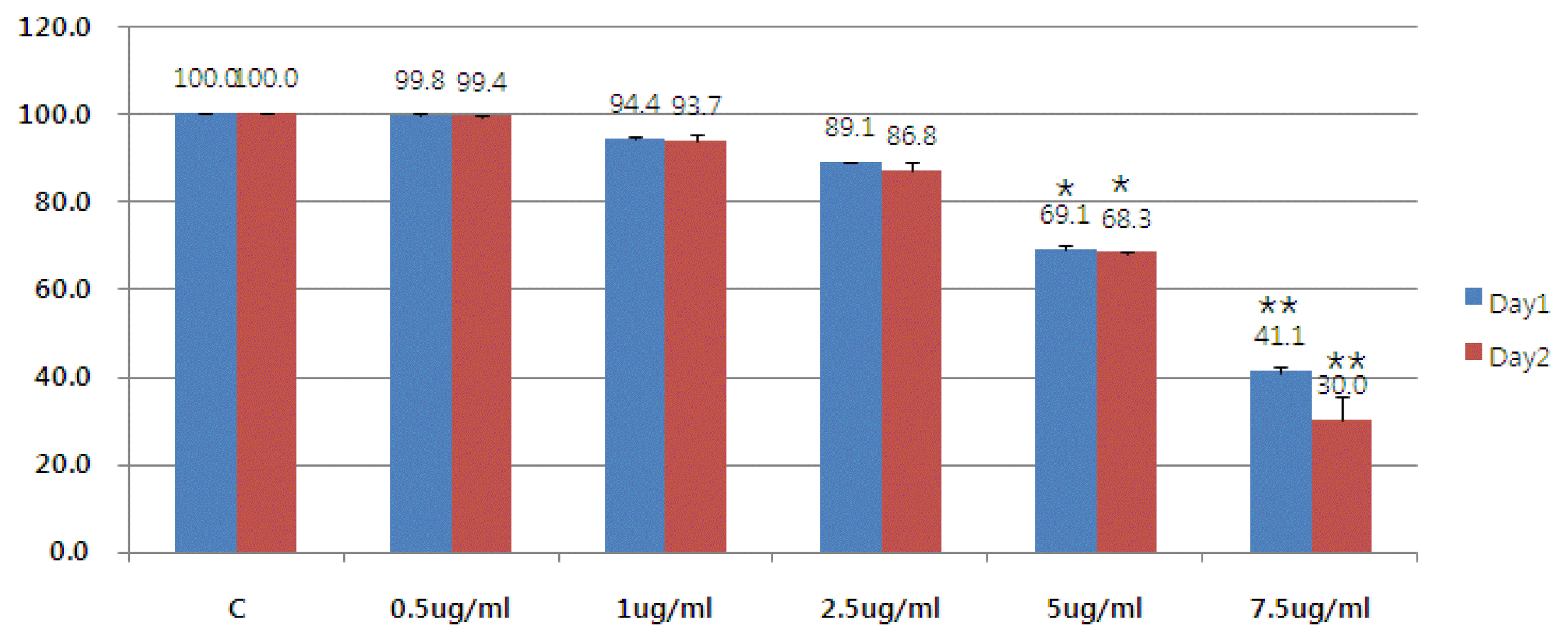
2. The Effect of Bovine (BV) on the Survival Rate of Liver Cancer Cells
Mistletoe used the previously well-known anti-cancer drug to compare and study the anti-cancer effect in liver cancer cells. The MTT method observed the effect on the survival rate of cells 24 hours and 48 hours after the poison was treated with liver cancer cells by concentration. Bong-dock significantly killed cells from a concentration of 5 ug/ml and at a concentration of 7.5 u/ml, more than 50% of the cells were killed (Figure 2).
3. Mystery mistletoe and Bongtoe are the two most successful liver cancer cells in Korea
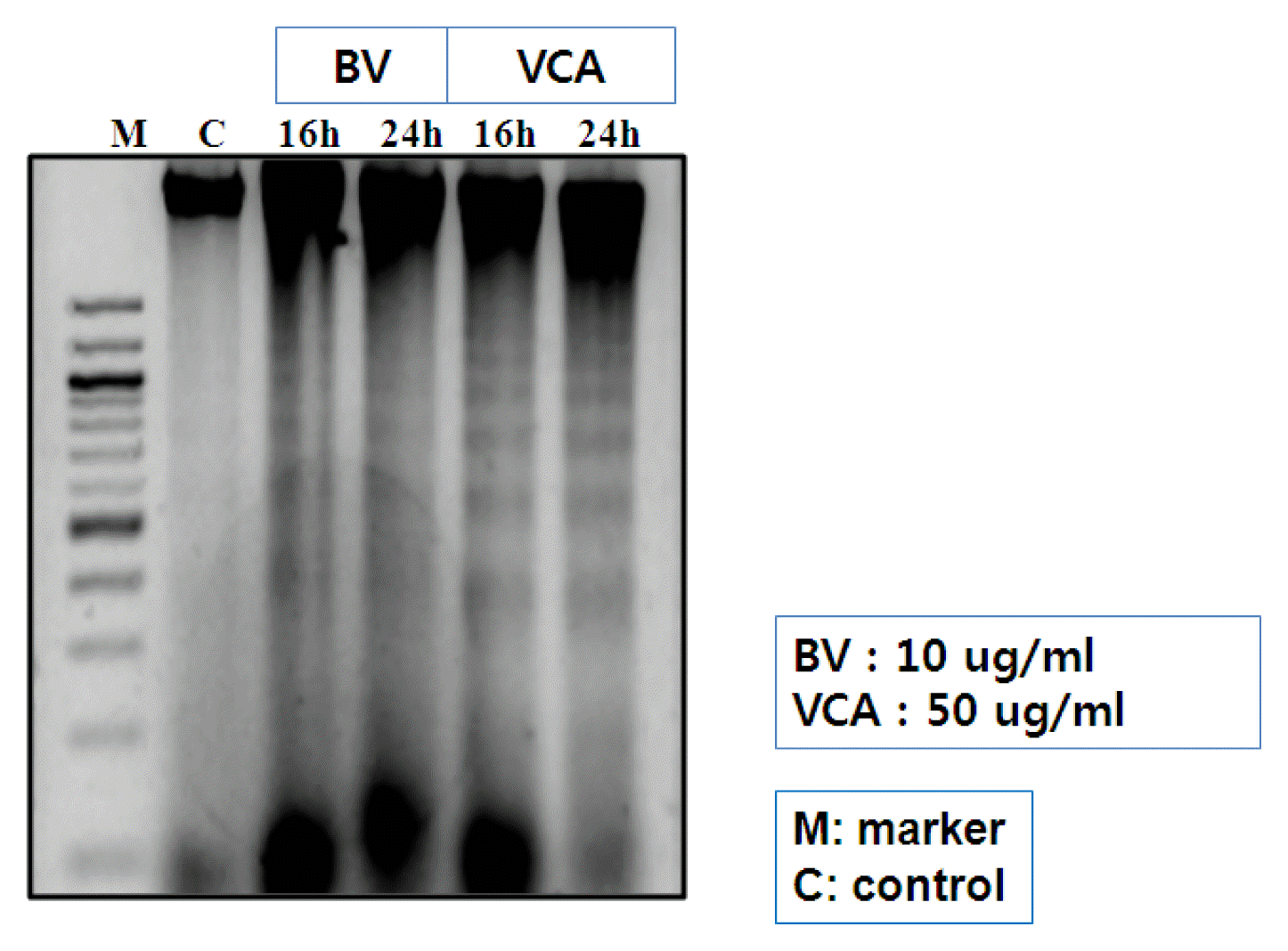
1) DNA fragmentation
The electroactivity was used to observe if chromosome DNA in the nucleus was cut to see if the apoptosis of the liver cancer cells in mistletoe and Bongto was achieved. We were able to see chromeoseome DNA cut into a ladder in 16 hours and 24 hours by Bong-dok and Mistleto (Figure 3).
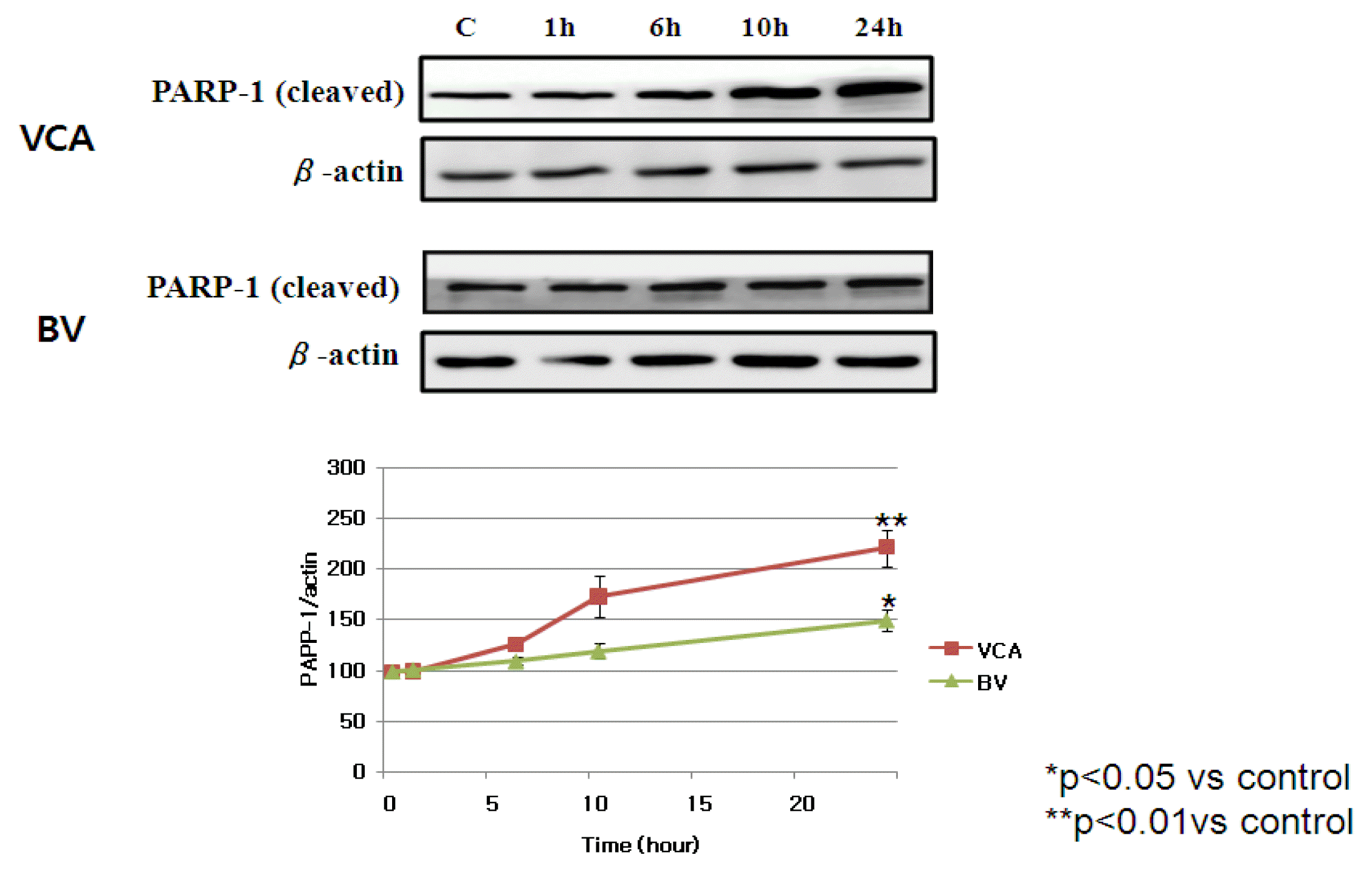
2) PARP-1 expression
Electrostatic activity was used to observe that the nucleus was cut off when mistletoe and poison induces apoptosis, and it was confirmed that the PARP-1 was activated. PARP-1 increased at activation by a small band of 29 kDa and observed that the cut band of PARP-1 by mistletoe and mutilation increased over time (Figure 4).
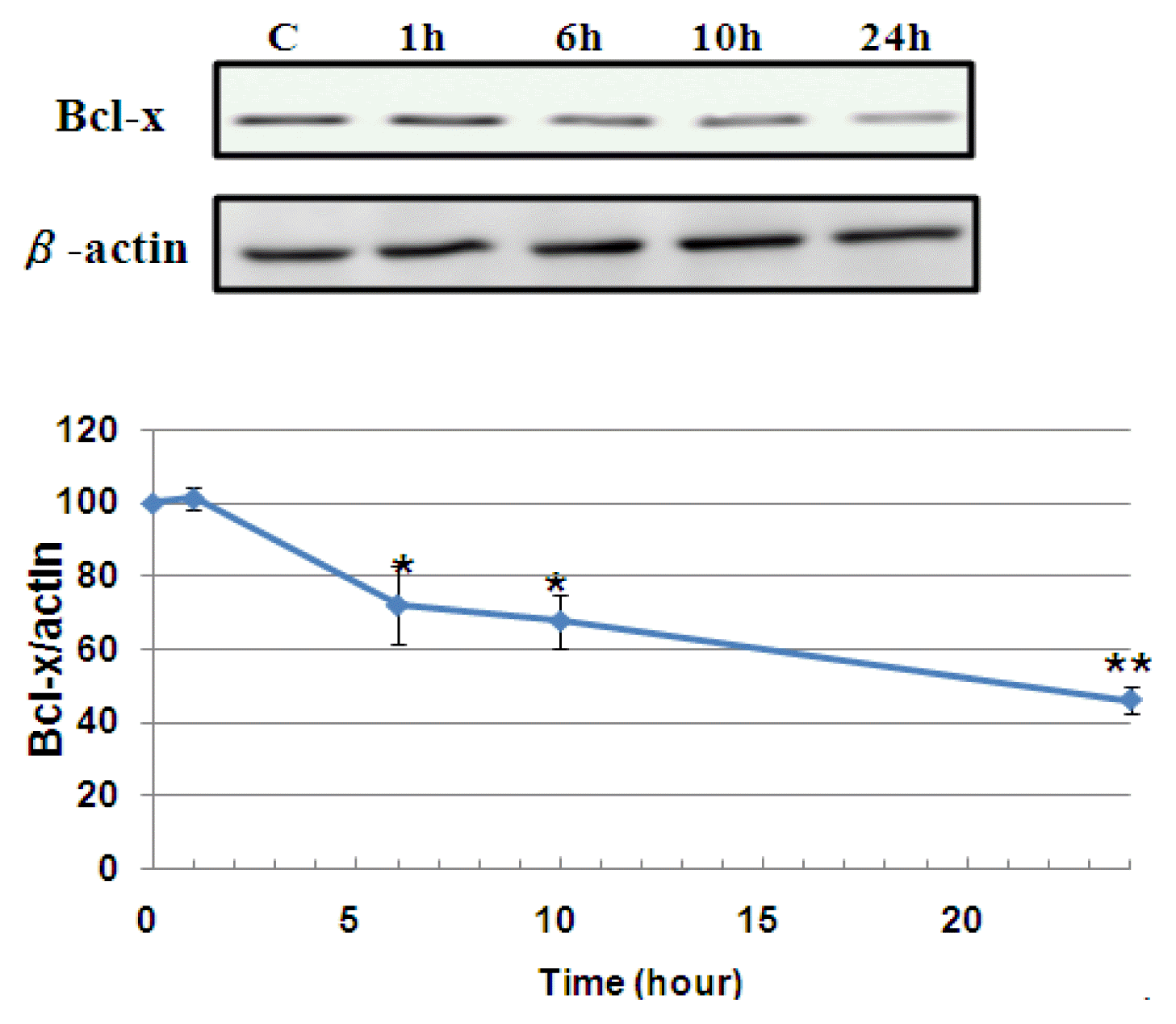
3) Bcl-x expression
Observing the accents of Bcl-x on a time-by-hour basis after mistreatment, mistletoe showed that only Mistletoe significantly decreased the amount of Bcl-x from six hours to 24 hours. These results suggest a different system of cell death signal transmission between Bong-dok and Mislotto (Figure 5).
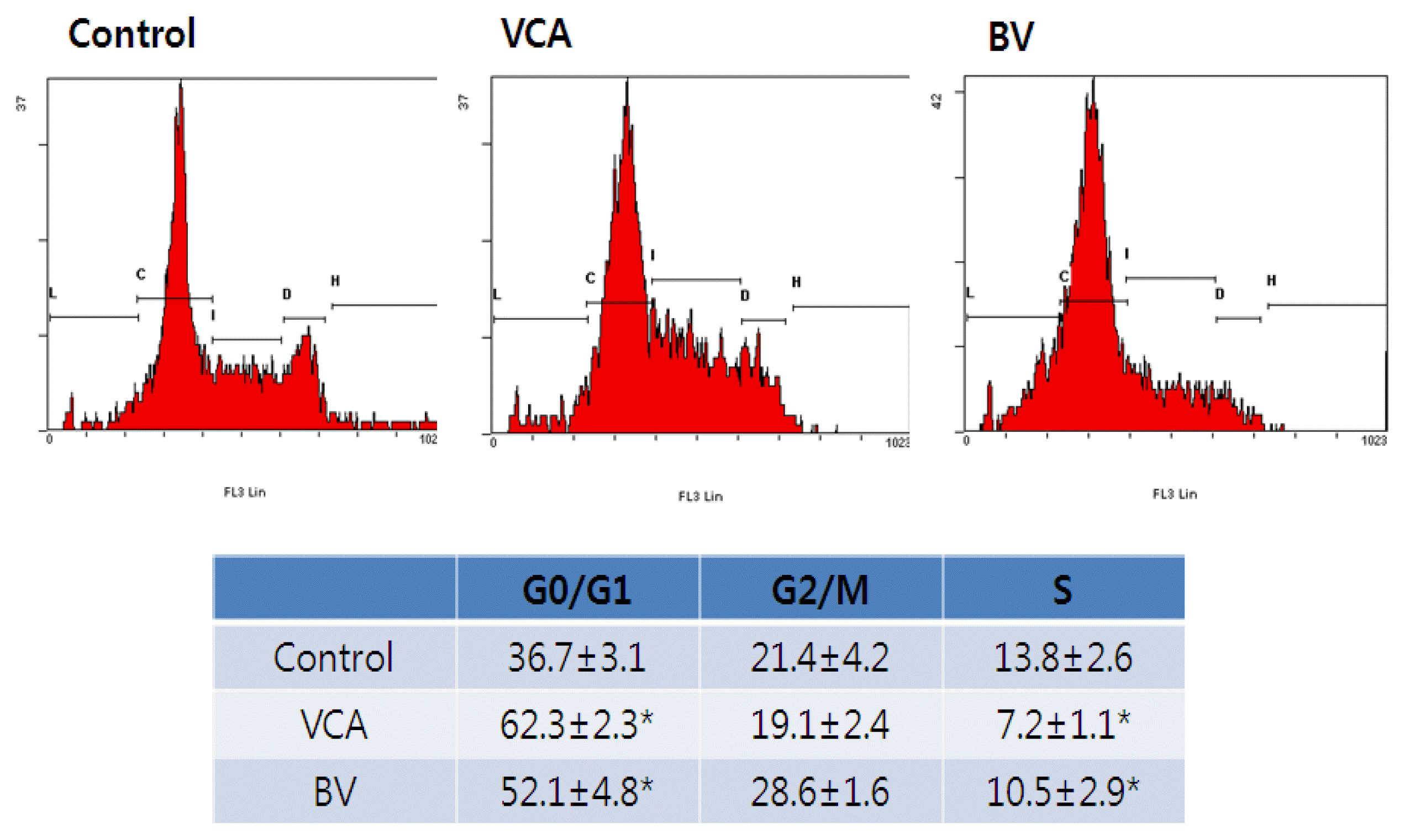
4) FACS analysis
After the treatment, cells were fixed at 9 hours and the nucleus was dyed to observe the distribution by cell cycle using flow chromometer. The number of cells in the G0/G1 phase has increased statistically significantly and the number of newly synthesized Sphases has also decreased by treatment of mistletoe and poison. These results suggest that mistletoe and bovine may also be partially involved in arresting the cell cycle of G0/G1 (Figure 6).
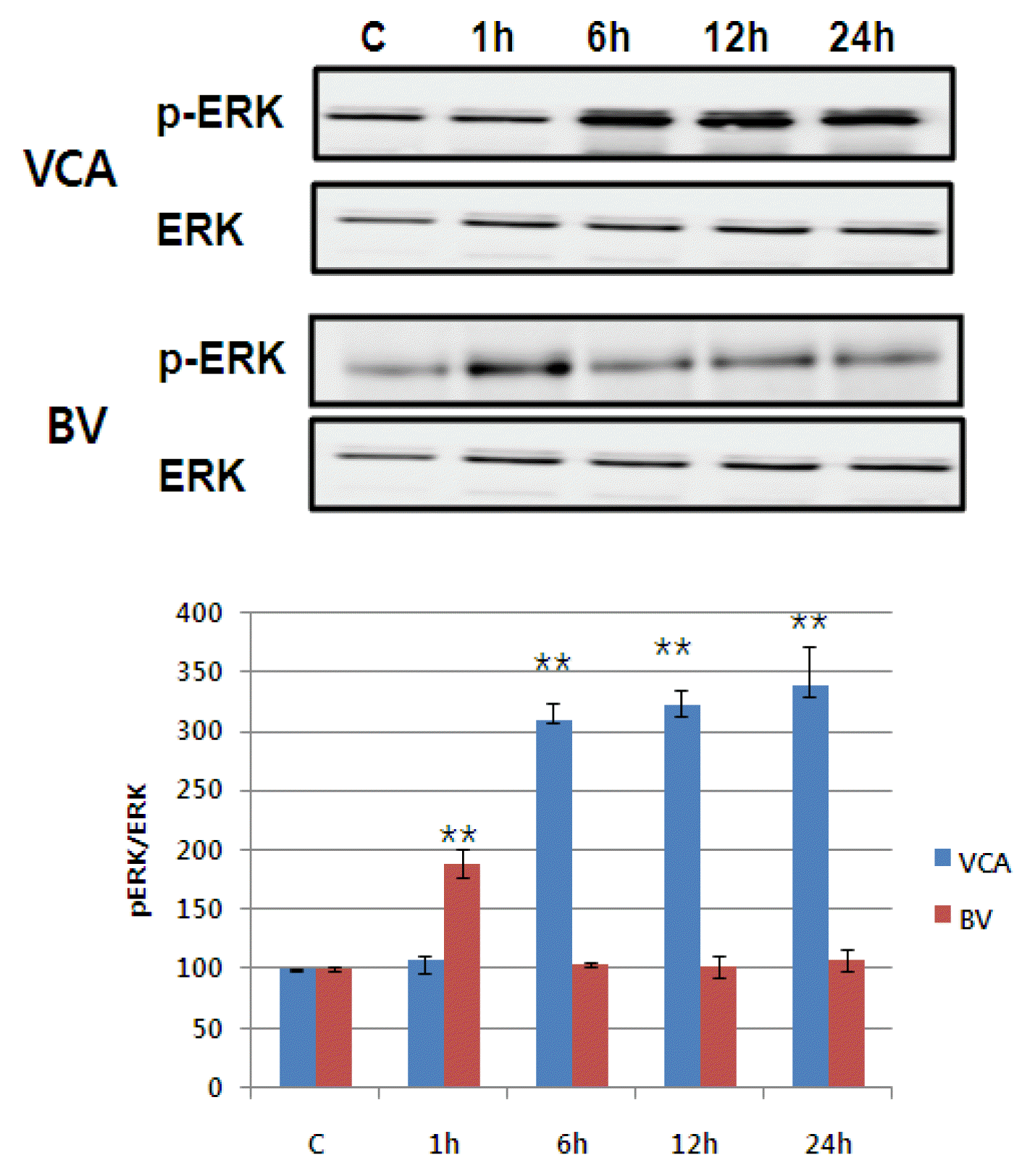
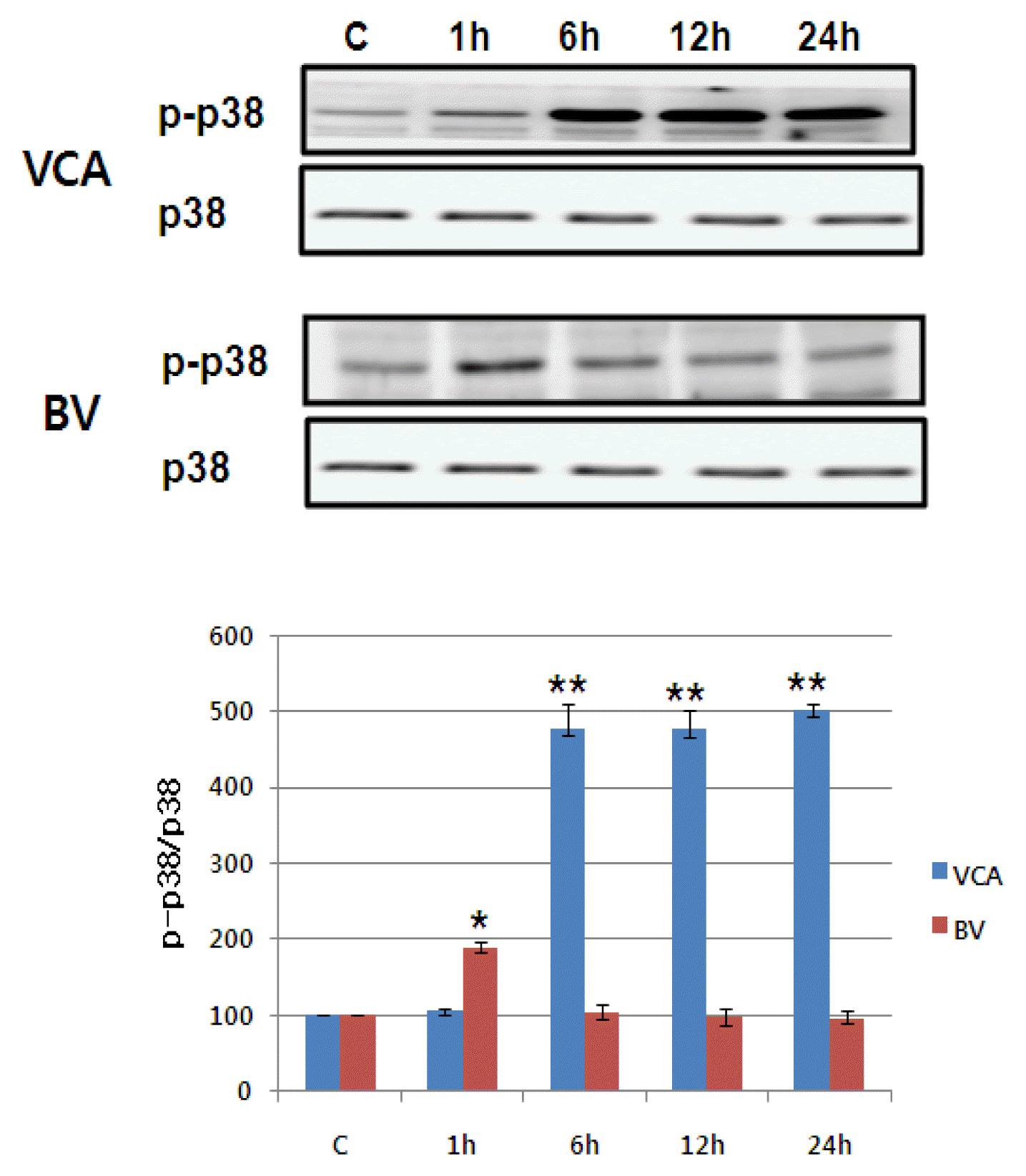
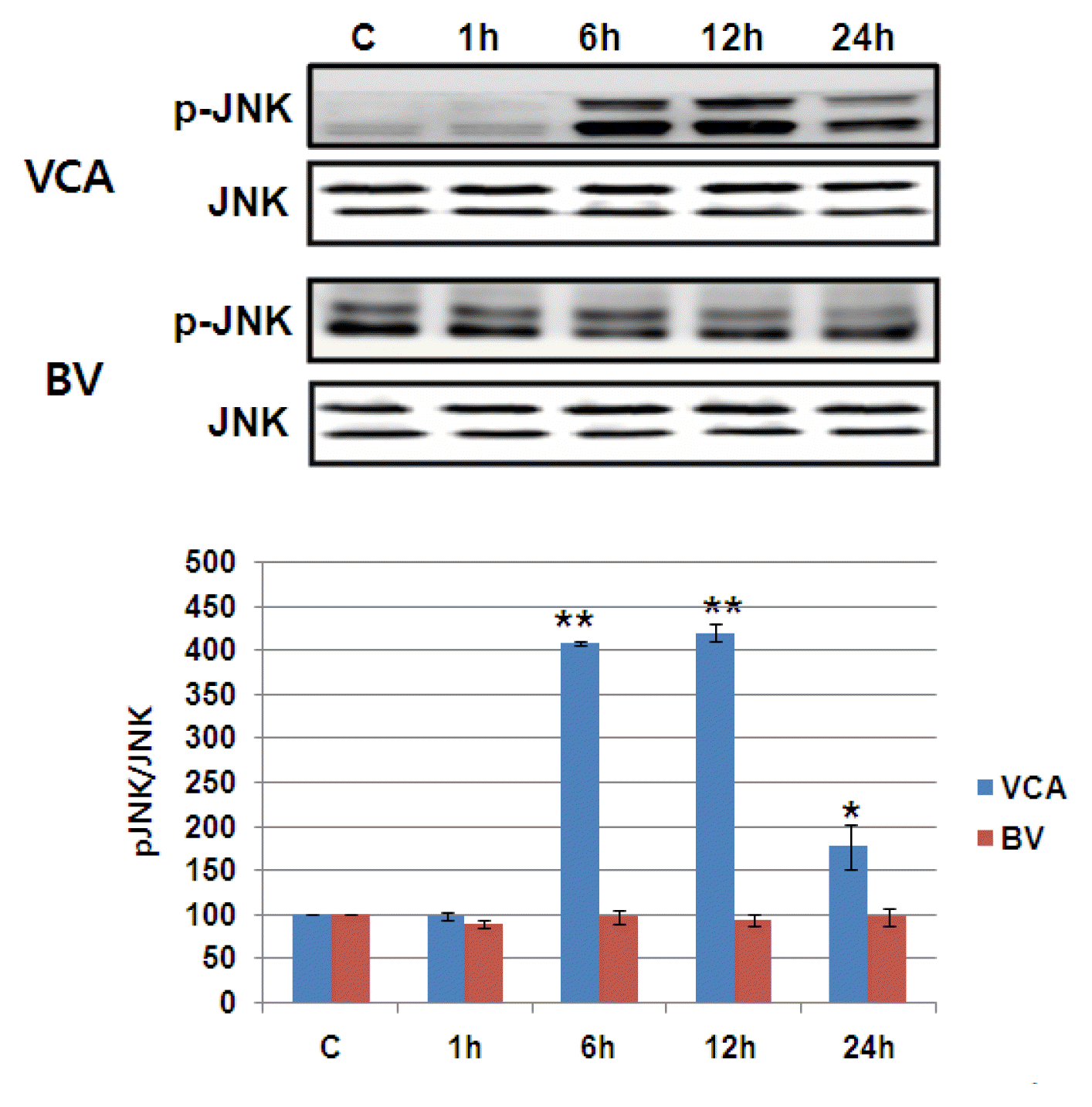
5) Activating MAPK family
The activity of MAPK/ERK, p38 MAPK, and JNK of the proteins reported to increase in activity in the event of mistletoe and bovine killing the liver cancer cells was observed using phosphorylation-specific antibodies6),7),8). Mistletoe continued for 24 hours from 6 hours after drug treatment, as phosphorylation of MAPK/ERK, p38 MAPK, and JNK began to increase three times more than that of controls. On the other hand, Bong-dok returned to the control level in six hours after the drug was processed, with the phosphorylation of MAPK/ERK and p38 MAPK doubling in just one hour. No activity of JNK was observed (Figure 7Fig. 8–9).
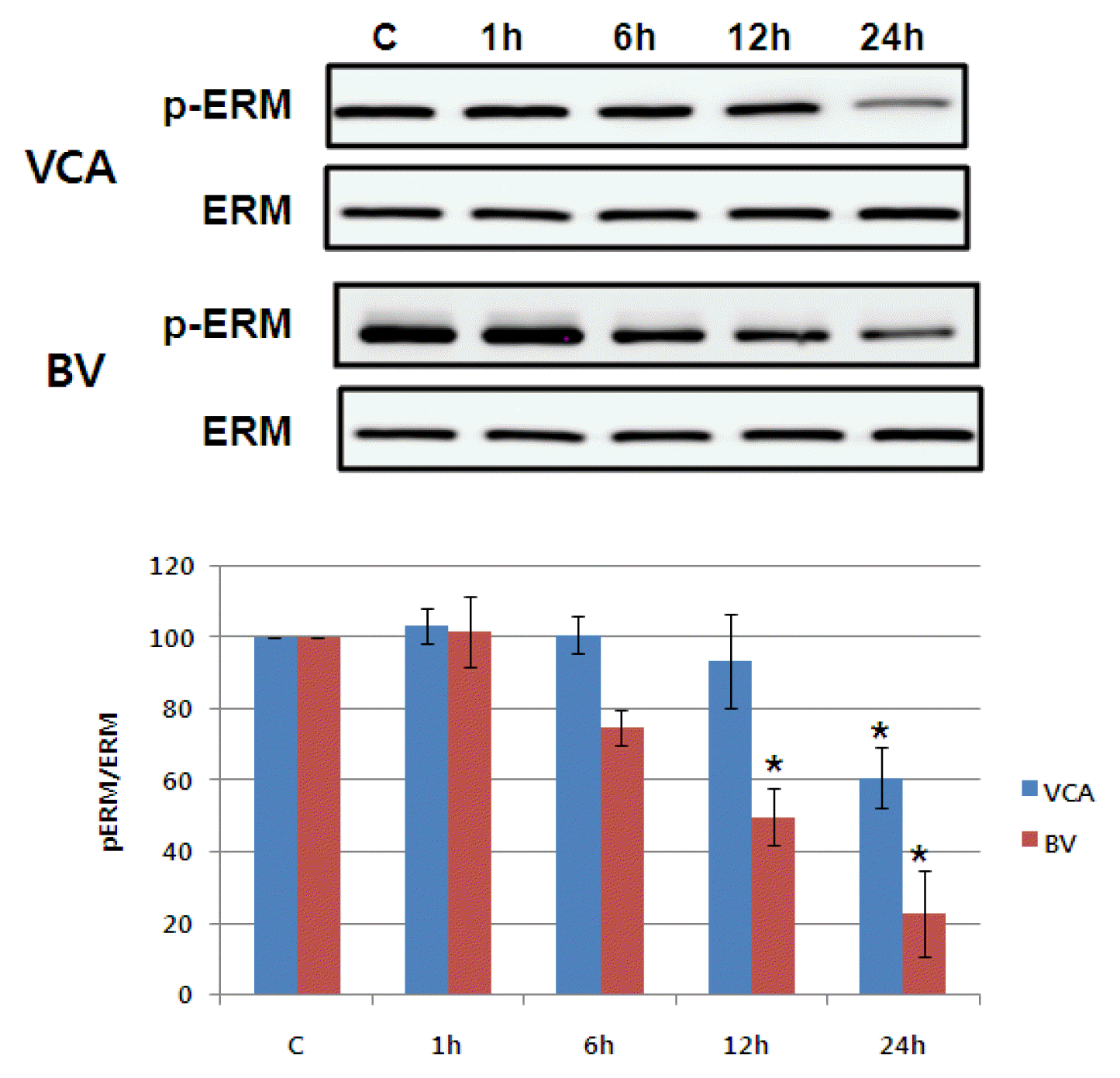
6) Activating ERM proteins family
The degree of activity of ERM protein, known to affect the cellular skeleton with another protein related to apoptosis, was performed as immunosuppression9). As shown in Figure 10, phosphorylation of ERM protein was reduced by mistletoe not more than 24 hours later. However, the number of bong poisons continued to decrease up to 24 hours, beginning with a six-hour decline. However, the amount of ERM protein has not changed, suggesting that only the phosphorylation of ERM proteins has decreased (Figure 10).
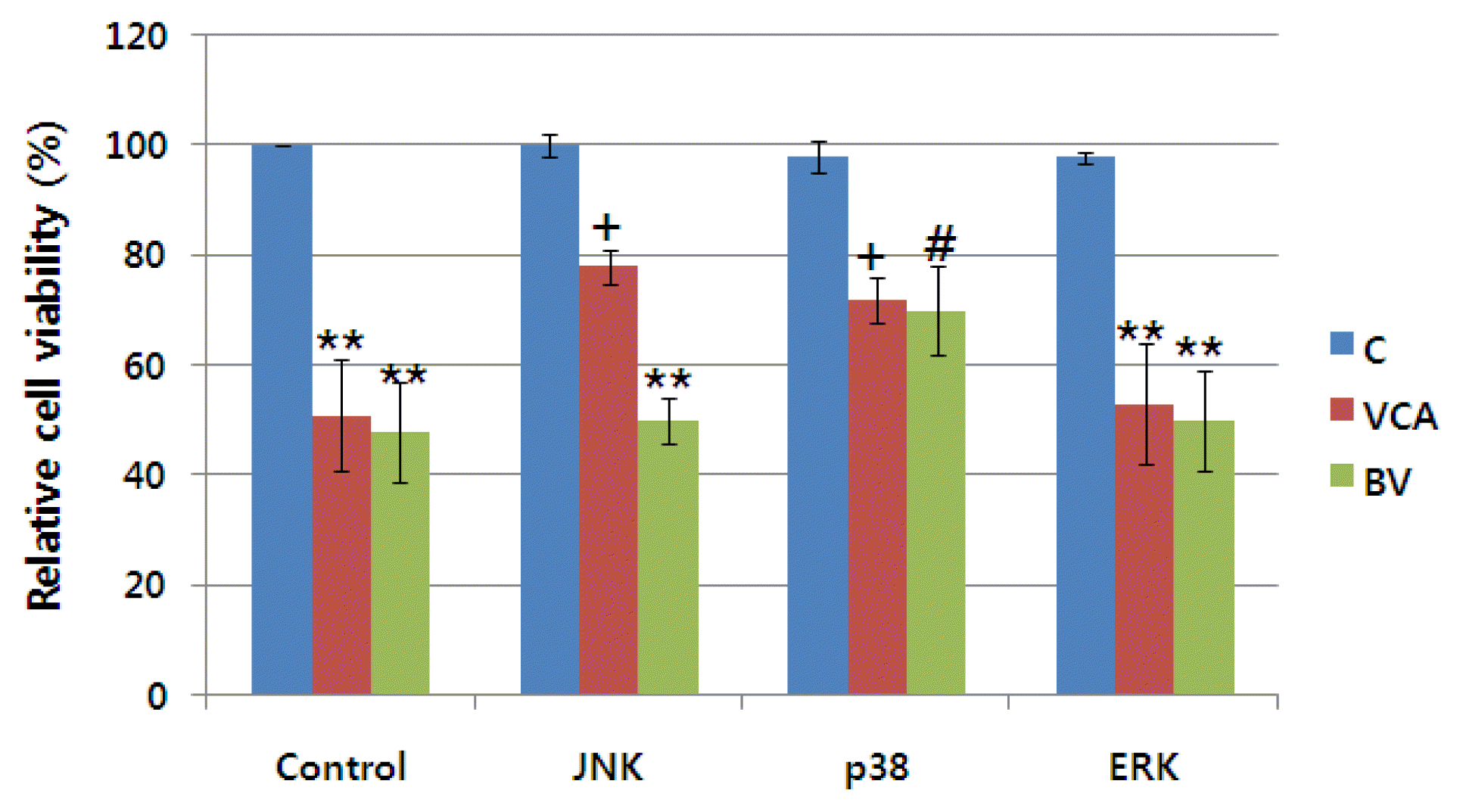
7) Identification of the signal transmission system for apoptosis of cells
We used active inhibitors of JNK, p38, and MAPK among the proteins that are known to be stressed or activated in apoptosis to identify the signal path of apoptosis by mistletoe and poison6),7),8),9). These inhibitors were given to the cells in advance, and mistletoe and poison were treated to perform MTT assay 24 hours after they were killed. As shown in Figure 11, the apoptosis of cells by mistleto was partially inhibited by JNK and p38 inhibitors. The cell killing effect of the poison was partially inhibited by the treatment of p38 inhibitors (Figure 11, 12).
Result & Discussion
In Korean medicine, mistletoe is known to be effective in controlling the central, diuretic, coercive, sedative, anti-cancer, and cardiovascular events.
The efficacy of mistletoe includes quercetin, quercitrin, aerialin, and oleanolic acid, among which quercetin induces immune suppression by reducing the number of white blood cells such as eosinophil and neutropyl, and oleanolic acid has been shown to be immune-boosting and anti-immune reactions.
There are reports that Mistletoe therapy uses different types of mistletoe to treat cancer10), and that the lectin-II component induces apoptosis for the human myeloid leukemia cells11). Immunodeficiency substances extracted from Visum album L. are applied to cancer and AIDS12). Fischer et al13) showed a sensitive response to the adjustment of T lymphocytes in CD4+ by processing mistletoe extract, while Park et al14) reported that the lectine component of mistletoe had an active lymphocytes effect, and Tasneem et al.15) reported that fermentation of mistletoe extract had an effect on inhibiting tumor cells.
Be Venom (BV) is a chemical that is highly likely to be characterized by both natural and hormonal agents and bio-therapeutics, and is a Pharmacology peptide components such as methylin, agolapin, MCDptide, etc., and enzymises (PLA2,alerton, GLA2, and GosThere is A2 et al.5),16).
MAPK (Mitogen-active proteinase), which is responsible for various functions such as cell proliferation, differentiation, and apoptosis, delivers extracellular stimuli to the nucleus from the membrane of cells to the receptor of growth hormones, cytokines, and stress de.
MARK can be classified as ERK (cell’s signal activation enzyme), JNK (c-JUN N-terminal active enzyme), p38 MAPK (ERK1/2) is primarily concerned with the signal transmission of growth hormones and is responsible for the growth and differentiation of cells, and the pKNAP, which are classified as stress-active enzymes, can be triggered by external pKNAPs.
In response, the author measured the degree of cell activity by concentration, time, and the activation of the protein involved in cell survival and apoptosis after administration of mistletoe extract and poison to Hep G2, a cell owner of liver cancer cells.
In general, in order to find efficacy for the development of new anti-cancer drugs or to find the sensitivity of the previously developed anti-cancer drug, it is necessary to examine the growth inhibition of tumor growth of the drugs outside of the living system prior to the application phase of animal testing. Several methods have been developed to measure the cell’s survival indirectly, since the number of samples is not available because it requires too much effort and time, even though a direct and ideal way of accurately measuring cells or living cells is to be treated with a trypan blueter in cells, and then count living cells using a microscope and a hemocytmeter. To this end, the in Vitro pharmacological search method of cancer drugs is widely used as a cytotoxic and cytotoxic search method by using 96-well plates and using ELISA reader to read many specimens quickly and objectively. MTT testing is used extensively for the purpose of primary screening tests on the sensitivity of cancer drugs, but can also be very useful for testing the sensitivity of cells to growth factors. In this experiment, the survival rate of cells by MTT test was determined by the concentration of mistletoe and poison and liver cancer cells dying. However, when comparing the two substances’ anticancer effects by dose, mistletoe twice as much as Bongto’s in order to achieve the same cellular toxicity effect 24 hours after treatment, but 48 hours later there were no differences in the effect of apoptosis of the two substances. These results show that the drug lasts a long time, with mistletoe dying over time compared to the poison
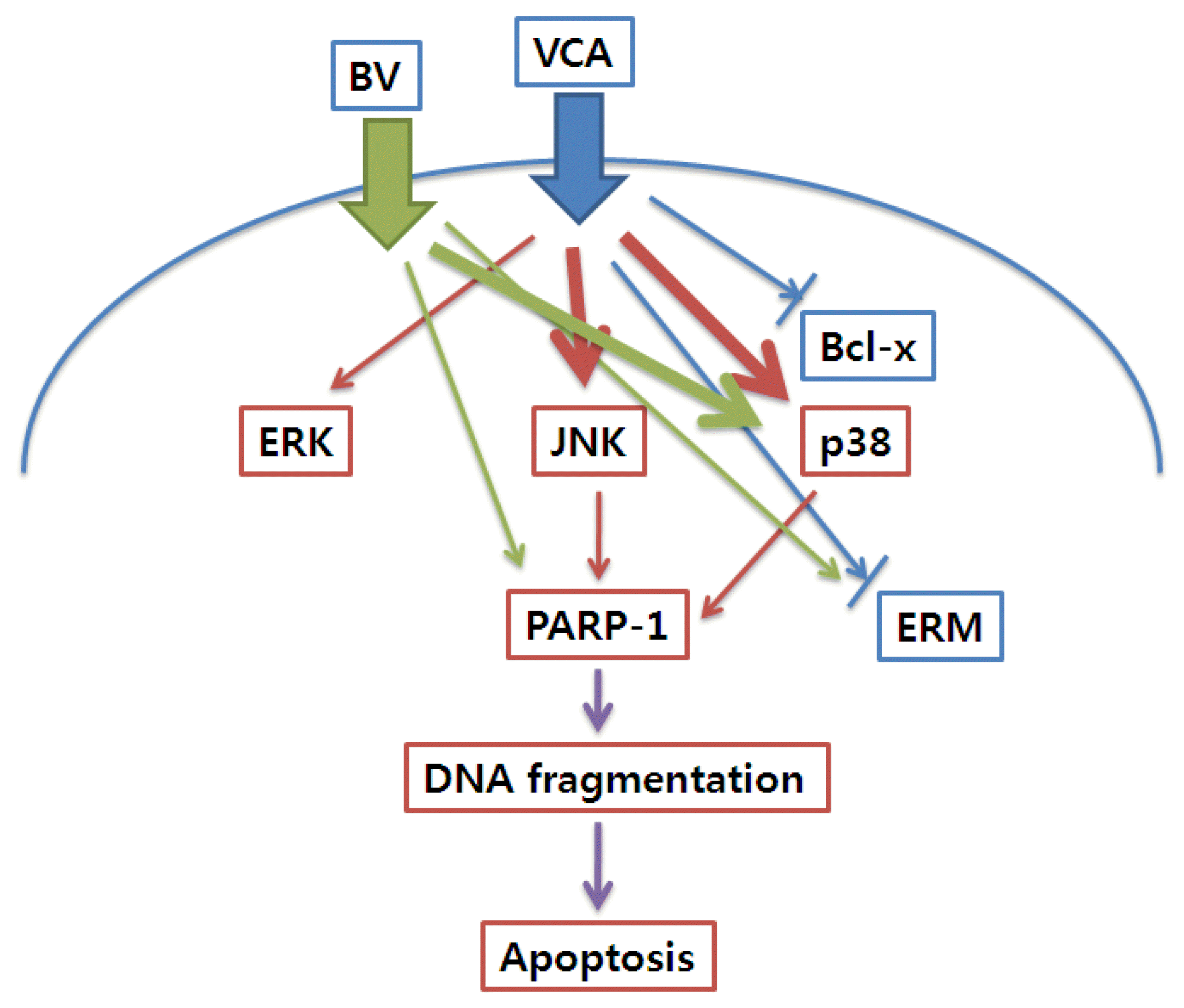
In general, if a cell’s death occurs due to toxicity, the cell’s death is caused by necrosis, inflammation, and cell death through one of the pathways. A necrosis is a group cell death that occurs quickly, and the characteristic of a collective cell death is that the membrane of the cell is destroyed first. But cell death, also known as apoptosis, is caused by the condensation and decay of the nucleus through various pathways inside the cell, and among the various cell death observed from the toxin, cell death caused mostly by the apoptosis. The most deeply studied biochemical event in Apoptosis is the amputation of the nuclear DNA double helix. These fragments appear in the form of ladders of DNA characterized by the agar-sagel electrophoresis 16). The protein involved in the cutting of these nuclear DNA double helix is known to be PARP-117),18). PARP-1 is a zinc dependent protein that is known to be activated when DNA strand breaks are triggered19), especially when cells die20). In this study, we found that DNA was cut into a ladder by mistletoe and poison, and we could observe the activated PARP-1. Therefore, these results suggest that mistletoe and bovine were caused by apoptosis, not necrosis, when they induced apoptosis. Typically, cell death apoptosis is done through a series of intracellular signalling systems, among which reducing the amount of protein that inhibits apoptosis also contributes to the cell death. A typical protein that suppresses these cell apoptosis is Bcl family protein21) Mistletoe began significantly reducing the expression of Bcl-x after six hours and then continued to decrease until 24 hours.
Observed by FACS as to which cell cycle was stopped by mistletoe and Bongto induces cell death, which led to cell proliferation and apoptosis, the number of cells in the G0/G1 phase increased statistically significantly and the number of newly synthesized spase was also reduced. These results suggest that mistletoe and bong poisons may also be partially involved in arresting the cell cycle of G0/G1. preparations for bee venom.
As this apoptosis progresses, a series of intracellular signalling systems are activated. MAPK family (MAPK/ERK, p38 MAPK, JNK) is known as their representative protein 6–8). JNK and p38 are known to be proteins activated by external stresses such as osmotic stress, UV irradiation, heat shock, oxidative stress, probabilistic cytokines22), p38 and JNK are known to be suppressed by external stresses that inherently inhibit the activity.23),24). Conversely, the MAPK family protein is known to be involved in cell survival or differentiation, which is controlled by the degree of activation of the MAPK family protein in the cell or by the balance of the period22). Persistent activation leads to apoptosis of cells, but temporary activation leads to cell survival or differentiation.22) This study shows that phosphorylation of MAPK family protein by mistleto has increased threefold in six hours after treatment to 24 hours. However, it only activated MAPK/ERK and p38 MAPK and doubled in just one hour after the poison was processed, recovering to the baseline in six hours. In addition, the apoptosis by mistletoe was partially recovered by treatment of JNK and p38 specific inhibitors, and the apoptosis by the poison was partially mitigated by p38 inhibitors. In the case of mistletoe, it is consistent with the existing results to demonstrate that the activity of JNK and p38 is sustained and strong enough to cause apoptosis of cells. However, unlike the existing results, the apoptosis of cells was partially inhibited by the treatment of p38 active inhibitors, despite the temporary activation of p38.
These results suggest that even a transient p38 activity can lead to partial apoptosis, suggesting the involvement of other intracellular signalling systems.
ERM proteins exist in inert state through in-molecular interaction within the cytoplasm, but when signals are transmitted from outside, the binding is released and activated to link cell membrane proteins with bone protein. Activation mechanisms are the phosphorylation of the Treonin remnants of the C-terminal. The upper control factor of ERM protein, identified so far, is Rho. The protein is known as an active-based cytoskeletal organization and is reported to have a direct impact on the activation of ERM proteins. Another upper element of ERM protein is known as Akt25). Akt protein is protein-serine/threonin case, PKB, whose function is known to play an important role in the development and progression of various cancer cells. The inhibition of ERM proteins is the dehumanization of the trenaline residue that was phosphated. This dehumanization is known to occur in the in vitro by myosin light chain phosphatase and protein phosphatase 2C, which occurs in the early stages of apoptosis with micro-fusion damage, which increases ERM protein’s movement to the cell vagina9). Akt’s dehumanization is known due to mistletoe’s anti-cancer mechanisms26). Therefore, Akt is likely to be involved in dehumanizing the ERM protein by mistleto.